Impact of SLCO1B3 Ser112Ala Genetic Variation on Efficacy and Safety of Paclitaxel in Iraqi Women with Breast Cancer
Download
Abstract
Objective: This study intended to detect genetic polymorphism in active transporter SLCO1B3 Ser112Ala (rs4149117) and study the effect of this variation on efficacy and safety of paclitaxel in Iraqi women with breast cancer.
Methods: The cross-sectional observational study was involved 150 women with breast cancer who received paclitaxel. These women were assessed individually in the 2nd week of paclitaxel therapy using a questionnaire to obtain demographic data, including age and body mass index, and the probability and severity of paclitaxel’s side effects. At the same time, neutrophilia count and breast cancer markers were assessed.
Result: The wild type (TT) was detected in about 14% of breast cancer cases. While the mutant type (GG) and the heterozygous type (TG) were detected in about 65% and 21% of the cases, respectively. The results did not exhibit a significant difference among Iraqi breast cancer women who carried TT, TG, and GG genotypes regarding breast cancer markers (CA 15.3 and CEA) and paclitaxel related adverse effects (neutropenia, oral mucositis, and peripheral neuropathy) at P > 0.05.
Conclusion: The SLCO1B3 334T>G genotyping was significantly distributed among Iraqi women with breast cancer. Still, there was no significant association between the SLCO1B3 334T>G genetic variation and efficacy and safety of paclitaxel.
Introduction
Breast cancer is the most common cancer occurring worldwide, accounting for 1:4 cases among women, and is the most commonly diagnosed cancer in the majority of countries [1]. It is a highly deadly cancer disease and comprises around 34.35% of total Iraqi women with cancer [2]. Breast cancer is known by [multifactorial, which is caused by heredity, environmental factors, the aging process, habit, and nutritional factors. Over the past 10–15 years, treatment concepts have evolved to account for this heterogeneity, with an emphasis on improving therapeutic benefits and reducing the adverse effects of chemotherapy [3, 4]. Breast cancer markers, including cancer antigen (CA15.3) and Carcinoembryonic antigen (CEA), are frequently used to identify the prognosis and to monitor the responsiveness of breast cancer to chemotherapy [5]. Paclitaxel is a chemotherapeutic agent in the taxane family that plays a pivotal role in the treatment of breast cancer, among other malignancies [6]. Although paclitaxel’s effectiveness in breast cancer in both early-stage and metastatic settings stems from its unique mechanism of action and broad anticancer activity [7], its resistance has been noted in some cases [8]. Several researchers studied the reasons for this resistance and demonstrated the roles of many factors, like the cytochrome P450 system inactivated paclitaxel [9], modification of cancer cell cycle regulation via overproduction of microtubulin-related proteins [10], and expression of ABC transporters [11]. Paclitaxel resistance remains a critical therapeutic problem because many women with breast cancer are on paclitaxel therapy. Another paclitaxel therapeutic–related problem is serious adverse effects, including neutropenia, peripheral neuropathy, alopecia, and oral mucositis [12] . The severity of these side effects may be consistent with decreased efficacy of paclitaxel. The explanation for this phenomenon remains unclear and requires further studies to identify the causes and overcome them [13]. The SLCO1B3 gene encodes the solute carrier organic anion transporter family member 1B3, commonly known as OATP1B3, a transmembrane transport protein that belongs to the organic anion-transporting polypeptide (OATP) superfamily. These transporters facilitate the uptake of a broad range of endogenous and exogenous organic anions into cells. OATP1B3 is predominantly expressed on the basolateral (sinusoidal) membrane of hepatocytes in the liver [14]. OATP1B3 mediates the liver uptake of many drugs, thereby influencing their pharmacokinetics, and its substrates include taxane, methotrexate, imatinib, statins, and other compounds [15]. Genetic variations in SLCO1B3 can lead to inter-individual variability in the function of the OATP1B3 transporter. Numerous single-nucleotide polymorphisms (SNPs) have been identified in the SLCO1B3 gene. Some are non-synonymous mutations resulting in amino acid changes that modify transporter activity [16]. Certain malignancies aberrantly express OATP1B3, which can have implications for tumor biology and drug delivery to tumors. SLCO1B3 Ser112Ala (rs4149117) genetic variation is relatively common across ethnicities and clinically associated with variability in drug levels, like in lung transplant patients on mycophenolate; those with GT or TT had a higher risk of chronic rejection than GG, suggesting a possible functional effect on drug disposition [17]. The Aim of this study was to detect genetic polymorphism in active transporter SLCO1B3 Ser112Ala (rs4149117) and study the effect of this variation on efficacy and safety of paclitaxel in Iraqi women with breast cancer.
Materials and Methods
Patients
Iraqi women age 50 – 78 years old with breast cancer were diagnosed according to the National Comprehensive Cancer Network (NCCN) clinical practice guideline [1]. These women were inpatients randomly recruited from the oncology department of Imam Al-Hussein Medical City after obtaining consent. Women were considered for inclusion in this study if they received paclitaxel as monotherapy at 80 mg/m², administered as a 1-hour intravenous infusion every week for at least two cycles, with no other concurrent diseases. Women with cardiovascular diseases, diabetes mellitus, hepatic dysfunction, hyperthyroidism, taking drugs that affect paclitaxel’s pharmacokinetic and pharmacodynamic properties, such as ketoconazole, erythromycin, rifampin, phenytoin, abaloparatide, abametapir, abatacept, and abciximab were excluded.
Determination of sample’s size
The number of women involved in this study was calculated using Fisher’s formula. The population size of the target category was knowing from the oncology department of Imam Al-Hussein Medical City which is about 242 women with breast cancer and received paclitaxel [18]..
N = (Z2PQ)/d2------------------------------------- [19]
Where N is the desired sample size and p is the prevalence and could consider p=50%=0.5 due to the expect proportion was obscured., whereas Q=1-p = 0.5, Z is the standard normal deviation of 1.96 at 95% CI, and D is an error agreeable limit of 0.05 at 95% CI. Thus,
N = ((1.96)2 X 0.5 X 0.5)/(0.05)2 = 384
The desired size could diminish using the reduction formula because the population size is < 10,000:
R = N/(1+ N⁄n)------------------- [18]
Where n is the population size of women involved in the study.
Then,
R = 384/(1+ 242) = 149.6
These 150 women were randomly selected.
Study design
The cross-sectional observational study was conducted from December 2024 to March 2025, which involved 150 women with breast cancer who received paclitaxel. These women were assessed individually in the 2nd week of paclitaxel therapy using a questionnaire to obtain demographic data, including age and body mass index (BMI), and the probability and severity of paclitaxel’s side effects. At the same time, neutrophilia count and breast cancer markers were assessed.
Blood collection
From each woman, approximately 6 mL of blood was withdrawn and divided into two parts: 2 mL was placed in an EDTA tube for the absolute neutrophil count (ANC) and genetic analysis, and 4 mL was placed in a gel tube to obtain serum for assessment of breast cancer markers.
Breast cancer markers measurement
The serum CA15.3 and CEA levels were measured using the chemiluminescent microparticle immunoassay technology [19] with the aid of ARCHITECT i1000SR immunoassay analyzer and CA15.3 and CEA kits (Abbott, USA).
White cell count
Differential complete blood count was carried out to assess hematological parameters, mainly the ANC, which is a critical indicator for evaluating bone marrow function and chemotherapy-induced myelosuppression in breast cancer patients receiving paclitaxel therapy. This test was performed by using an automated Swelab Alfa Hematology Analyzer (Boule Diagnostics AB, Sweden).
Assessment the severity of paclitaxel-induced neutropenia
The severity of paclitaxel-induced neutropenia was commonly graded by a scale that was derived from the Common Toxicity Criteria of the National Cancer Institute. This scale schemed neutropenia into four scores based on the ANC: score 1, ANC of ≥1. 5 to <2×109/L; score 2, ANC of ≥1.0 to <1.5×109/L, score 3, ANC of ≥0.5 to <1.0×109 /L, and score 4, ANC <0.5×109/L [20].
Assessment of paclitaxel-induced oral mucositis
The paclitaxel-induced oral mucositis was assessed according to the World Health Organization (WHO) scale that combines both clinical and functional/symptoms-based examination of oral mucositis. This scale was delineated oral mucositis as follows: score 0: No oral mucositis, score 1: erythema and soreness, score 2: ulcers, able to eat solids, score 3: ulcers, requires a liquid diet, and score 4: ulcers, alimentation not possible [21].
Assessment of paclitaxel-induced peripheral neuropathy
The peripheral neuropathy induced by paclitaxel was evaluated grounded on the WHO Common Toxicity Criteria for Peripheral Neuropathy that rating scale was pointed into 5 scores: score 0: no symptoms of neuropathy, score 1: paresthesia (a tingling, tickling or prickling sensation) and/or limited tendon reflexes, score 2: severe paresthesia and/or mild weakness, score 3: unendurable paresthesia and/or marked motor loss, and grade 4: paralysis [22].
Genotyping
Genomic DNA was isolated from whole blood samples utilizing the AddPrep™ Genomic DNA Extraction Kit (AddBio, Korea) in accordance with the manufacturer’s guidelines. The allele-specific PCR (AS-PCR) method was utilized to identify SLCO1B3 c.334T>G (rs4149117) gene polymorphisms, employing the Mastercycler Gradient thermal cycler (Eppendorf, Germany). This method is extensively employed for SNP genotyping owing to its superior specificity and economic efficiency in differentiating alleles based on single-nucleotide variations [23]. For each SNP, three allele-specific forward primers (one for each allele) and one common reverse primer were built according to Dr. Hassan Abo Almaali. The primer sequences for this gene are forward (Allele T) 5’-GGAACTGGAAGTATTTTGACAT-3’, forward (Allele C) 5’-GGAACTGGAAGTATTTTGACAC-3’, F o r w a r d ( A l l e l e G ) 5’-GGAACTGGAAGTATTTTGACAG-3’ and reverse 5’-CAGGTGAAGTTGTGAAGCC-3’, with a product size is 400 bp. Each PCR reaction was conducted in a total volume of 25 µL, comprising 3 µL of DNA template, 2 µL of each forward and reverse primer, 5 µL of 5× Master Mix (AddBio, Korea), and 13 µL of distilled water. The PCR parameters for rs4149117 included an initial denaturation at 95 °C for 5 minutes, pursed by 35 cycles comprising 25 seconds at 95 °C, 25 seconds at 55 °C for annealing, and 1 minute at 72 °C, culminating in a final extension at 72 °C for 5 minutes. After amplification, 5 µL of each PCR product was combined with 3 µL of loading dye and applied to a 1.5% agarose gel prepared with 1× TBE buffer and 0.5 µg/mL ethidium bromide. Electrophoresis was conducted at 100 volts for a duration of 35 minutes. The DNA bands were observed under UV light with a transilluminator and recorded using a digital camera. Band sizes were determined by comparison with a 100–1000 bp DNA ladder. All outcomes were validated by two independent evaluators.
Statistical analysis
The Statistical Package for the Social Sciences (SPSS 26) was used to perform statistical analysis. The Shapiro–Wilk test was used to investigate the normality of data distribution. Numerical data were descriptively presented as the mean and standard error of the mean (Mean ± S.E.) for normally distributed data and the median and interquartile range (Median ± IQR) for non-normally distributed data. At the same time, the non-numerical data were presented as numbers and %. Numerically normally distributed data, including age and BMI, were analyzed using the one-way ANOVA-Post Hoc-LSD, and numerically non-normally distributed data by using nonparametric tests and legacy dialogs – Kruskal-Walli’s test. Non-numerical data, including paclitaxel-related adverse effects, were analyzed by using the Chi-square test. The allele distribution of SLCO1B3 Ser112Ala genes was analyzed using the Hardy-Weinberg equation and nonparametric tests, legacy dialogs, Chi-square test. The P values less than 0.05 were considered statistically significant.
Results
Demographic data of breast cancer women
The demographic data of breast cancer women were investigated and the mean age and BMI were 52.02 years ± 0.87 and 29.36 mg/h2 ± 0.42, respectively.
The serum level of breast cancer markers
The serum levels of breast cancer markers were measured and the median of CA 15.3 and CEA were about 17.95 and 2.37 respectively.
The incidence and severity of paclitaxel related adverse effects
The paclitaxel-induced neutropenia was observed in all breast cancer women with different severity and score 3 was predominated. Most women with breast cancer treated with paclitaxel suffered from mucosa erythema, soreness, and difficulty eating, and scores 1 and 2 were highly incident among those patients. Paresthesia, weakness, and reduced tendon reflexes were common symptoms of peripheral neuropathy, which were observed in women with breast cancer who were treated with paclitaxel, and scores 1 and 2 were commonly diagnosed in those patients as presented in Table 1.
| Paclitaxel-related Adverse effects | No (%) | |
| Neutropenia | Score 1 | 25 (16.6) |
| Score 2 | 24 (16) | |
| Score 3 | 82 (54.7) | |
| Score 4 | 19 (12.7) | |
| Oral mucositis | Score 0 | 32 (21.4) |
| Score 1 | 50 (33.3) | |
| Score 2 | 59 (39.3) | |
| Score 3 | 9 (6) | |
| Score 4 | 0 (0) | |
| Peripheral neuropathy | Score 0 | 9 (6) |
| Score 1 | 70 (46.7) | |
| Score 2 | 55 (36.7) | |
| Score 3 | 13 (8.6) | |
| Score 4 | 3 (2) |
† Data expressed as No (%)
Genotyping
Prevalence and
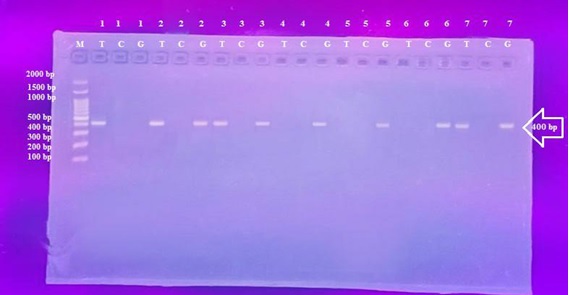
The clear bands with molecular size 400 bp represented the results of SLCO1B3 C334T>G (rs4149117) genotyping. The size of this gene amplicon was determined by comparing with a DNA ladder 100 - 1000 bp, genotype of which were classified into three genotypes including the major wild type (TT) for the allele T, the homozygous mutant type (GG) for the allele G, and the heterozygous (TG). as exhibited in Figure 1.
Figure 1. Genotyping of SLCO1B3 C334T>G (rs4149117) gene.
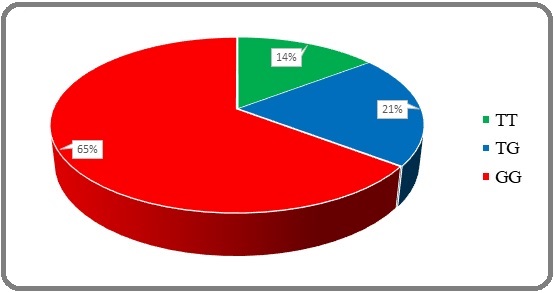
The wild type (TT) was detected in about 14% of breast cancer cases. While the mutant type (GG) and the heterozygous type (TG) were detected in about 65% and 21% of the cases, respectively, as presented in Figure 2.
Figure 2. The Prevalence of SLCO1B3 334T>G (rs4149117) Genotypes among Breast Cancer Women.
The allele and genotype distribution of SLCO1B3 C334T>G (rs4149117) are demonstrated in Table 2.
| Genotype (N:150) | Frequency (%) | Allele | Frequency | Chi-square value | P-value |
| TT (wild type) | 22 (14.67%) | T | 0.25 | 30.225 | 0.000000027 |
| TG (heterozygous type) | 31 (20.66%) | G | 0.75 | ||
| GG (homozygous type) | 97 (64.67%) |
† Hardy-Weinberg equation and Chi-Square, two sided P.value<0.05. N: number of study cases.
Effects of SLCO1B3 334T>G genetic variation on the breast cancer markers
The serum levels of breast cancer markers, including CA 15.3 and CEA, were measured to investigate the effect of different SLCO1B3 334T>G genotypes on the paclitaxel efficacy. The results did not exhibit a significant difference among Iraqi breast cancer women who carried TT, TG, and GG genotypes at P > 0.05, as shown in Table 3.
| Parameters | Alleles of SLCO1B3 334T>G Genotypes | P – value | ||
| TT | TG | GG | ||
| CA 15.3 (mIU /ml) | 16.6 ± 23.14 | 18.6 ± 18.2 | 18.5 ± 17.75 | 0.948 |
| CEA (mIU /ml) | 2.66 ± 8.18 | 2.07 ± 3.23 | 2.51 ± 2.14 | 0.445 |
† Kruskal-Walli’s, two sided P.value<0.05, Data expressed as median ± IQR
Effects of SLCO1B3 334T>G genetic variation on paclitaxel-related adverse effects
The high incidence of paclitaxel-related adverse effects, including neutropenia, oral mucositis, and peripheral neuropathy, was assessed to identify the possible impacts of different SLCO1B3 334T>G genotypes on the safety of paclitaxel. The results did not show a significant association between the SLCO1B3 334T>G genetic polymorphism and severity of the paclitaxel-related adverse effects (neutropenia, oral mucositis, and peripheral neuropathy) at P > 0.05, as explained in Table 4.
| Adverse effects | Alleles of SLCO1B3 334T>G Genotypes | P – value | |||
| TT | TG | GG | |||
| Neutropenia (%) | Score 1 | 2 (9.1) | 2 (6.5) | 21 (21.6) | 0.425 |
| Score 2 | 4 (18.2) | 5 (16.1) | 15 (15.5) | ||
| Score 3 | 13 (59.1) | 18 (58.1) | 51 (52.6) | ||
| Score 4 | 3 (13.6) | 6 (19.4) | 10 (10.3) | ||
| Oral mucositis (%) | Score 0 | 4 (18.2) | 6 (19.4) | 22 (22.7) | 0.917 |
| Score 1 | 9 (40.9) | 10 (32.3) | 31 (32) | ||
| Score 2 | 7 (31.8) | 14 (45.2) | 38 (39.2) | ||
| Score 3 | 2 (9.16) | 1 (3.2) | 6 (6.2) | ||
| Score 4 | 0 (0) | 0 (0) | 0 (0) | ||
| Peripheral neuropathy (%) | Score 0 | 2 (9.1) | 2 (6.5) | 5 (5.2) | 0.964 |
| Score 1 | 9 (40.9) | 15 (48.4) | 46 (47.4) | ||
| Score 2 | 8 (36.4) | 10 (32.3) | 37 (38.1) | ||
| Score 3 | 3 (13.6) | 3 (9.7) | 7 (7.2) | ||
| Score 4 | 0 (0) | 1 (3.2) | 2 (2.1) |
† Chi-Square, two sided P.value<0.05, Data expressed as No (%)
Discussion
Breast cancer is one of the most common spread cancers in Iraqi women, and the critical point for its management and preventing metastasis is selecting effective and safe chemotherapy [24]. Paclitaxel is a highly prescribed chemotherapy for the management of breast cancers with majestic effectiveness, but many cases developed resistance to it [25]. There are several factors believed to play roles in decreasing the efficacy of paclitaxel and producing poor clinical response. Some study mentioned that pathway impairment, epigenetic alteration, and genetic variation may be commonly included causing factors [26-28]. OATP1B3 is a highly expressed transporter that is encoded by the SLCO1B3 gene. It is responsible for the hepatic basolateral uptake of anion substrates; therefore, it plays a crucial role in the systemic elimination of drugs and toxins. It may be one of these factors that affects the response to paclitaxel [29].
The study showed that the polymorphism of the SLCO1B3 334T>G gene was present in Iraqi women with breast cancer, and the homozygous GG genotype predominated. The minor allele frequency of the G allele is significantly high (about 75%) in comparison to the reference major allele (T) in selected women. The study observed a significant variation in allele frequency of the SLCO1B3 334T>G among ethnicities, with low frequency in African American and Ghanaian subjects, in contrast to Caucasian, Han Chinese, and Mexican subjects [30]. Tsujimoto et al. mentioned that the genetic variation of SLCO1B3 334T>G is total connection imbalance in 79 Japanese individuals and the variant allele is highly frequency (72.8%) [31]. In Asian countries, the SLCO1B3 334T>G gene exhibited greater polymorphism and surprising interracial diversity among the Malay, Indian, and Chinese individuals, suggesting that this polymorphism may affect the pharmacokinetic properties, including the intracellular or extracellular transport of many substrates. Taxane chemotherapy may be believed to be one of them [32].
In this study, the serum levels of breast cancer markers (CA 15.3 and CEA) were adjusted within normal values in most Iraqi women with breast cancer. This revealed that paclitaxel effectively prohibited the metastasis. CA 15.3 and CEA were frequently performed for investigating, treatment monitoring, and advancing breast cancer. If the result value of CA 15.3 and CEA are ≥ 20 U/ml and 3 μg/L, respectively, this cancer is poorly responsive to therapy [33]. The study demonstrated that the SLCO1B3 334T>G genetic polymorphism did not play an important role in the effectiveness of paclitaxel against breast cancer in Iraqi women. These results are consistent with some other studies [30,34] . The relationship between the SLCO1B3 334T>G genetic variation and paclitaxel pharmacokinetic was investigated in five ethnic population (European Caucasian, American Caucasian, African-American, Mexican, and Han Chinese) and revealed that the plasma concentration of unbound paclitaxel was not significantly associated with the polymorphism of SLCO1B3 334T>G gene due to this study exhibited statistically insignificant associations between SLCO1B3 genotype and paclitaxel-pharmacokinetic elements including dose-timing area under the curve, peak plasma concentration, volume of distribution, and drug half-life [31]. A study done on patients of Asian race who received docetaxel as chemotherapy demonstrated that the polymorphism of the SLCO1B3 gene did not significantly alter the pharmacokinetic characteristics of docetaxel; therefore, it did not affect drug efficacy [35].
The results of this study showed an insignificant association between the SLCO1B3 334T>G genetic polymorphism and the severity of the paclitaxel-related adverse effects. In Spanish patients, the study demonstrated no considerable link between SLCO1B3 genotyping and paclitaxel-induced peripheral neuropathy [36]. Although paclitaxel induced many adverse effects, neutropenia, oral mucositis, nausea and vomiting, alopecia, and peripheral neuropathy are the most common adverse one [37-39]. A Japanese study investigated the role of SLCO1B3 334T>G genetic polymorphism in docetaxel-induced neutropenia, revealing that there were no significant associations between docetaxel therapy and incidence of neutropenia [40]. Paclitaxel-induced hematological toxicity (mainly neutropenia and thrombocytopenia) is a dose-related adverse effect, and accordingly, OATP1B3 is responsible for the transport of paclitaxel in hepatic metabolism. But the plasma concentration of unbound paclitaxel was not significantly associated with the presence of SLCO1B3 334T>G genetic variation. However, patients with one or two T alleles of the SLCO1B3-rs4149117 polymorphism exhibited a 19% lower sensitivity to thrombocytopenia but not neutropenia compared to those with homozygosity for the G allele (p = 0.00279) [41].
In conclusion, this study concluded that the SLCO1B3 334T>G genotyping was significantly distributed among Iraqi women with breast cancer. Still, there was no significant association between the SLCO1B3 334T>G genetic variation and efficacy and safety of paclitaxel.
Acknowledgements
General
We thank all department members of Oncology Department of Imam Al Hussein Hospital, including nurses, service workers, resident doctors, and statistics employees. We would like to acknowledge our institutional affiliations for providing the necessary infrastructure and resources to complete this work, but no direct financial support was received.
Funding Statement
There is no support in financial, but it is Self-financing
Approval
This study was approved by the scientific and ethical committee at Kerbala University, College of Pharmacy with the project being assigned No: 2022HU9 and by Ministry of Health of Iraq - Kerbala health department with the project being assigned No: 433.
Conflict of Interest
The authors declare that they have no conflicts of interest
Ethical Declaration
The scientific and ethical committee at Kerbala University, College of Pharmacy.
Authors Contribution
A.B. participated to the study’s design and execution,analysis of the results, and composition of the manuscript.
A.A. and A. H. contributed to the review and editing of the study and supervised the research. A. S. contributed to data collection and critically appraised the manuscript. All authors have critically reviewed and approved the final draft and are responsible for the content and similarity index of the manuscript.
References
- Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL , Soerjomataram I, Jemal A. CA: a cancer journal for clinicians.2024;74(3). CrossRef
- Cancer in Iraq, general view of annual report 2022 Salih HH , Abd SY , Al-Kaseer E, Al-Diwan J. Journal of Contemporary Medical Sciences.2025;10(6):475-477. CrossRef
- [Retracted] Global Increase in Breast Cancer Incidence: Risk Factors and Preventive Measures Kashyap D, Pal D, Sharma R, Garg VK , Goel N, Koundal D, et al . BioMed Research International.2022;2022(1). CrossRef
- Molecular Docking Study of Five Novel 1,2,3-Triazole Linked Metronidazole Derivatives as Cytotoxic Agents Sayad HHA , Sahib AAAA , Oleiwi ZK . Karbala Journal of Pharmaceutical Sciences.2024;15(24). CrossRef
- Diagnostic impact of CEA and CA 15-3 on chemotherapy monitoring of breast cancer patients Hasan D. Journal of Circulating Biomarkers.2022;11. CrossRef
- Paclitaxel: Application in Modern Oncology and Nanomedicine-Based Cancer Therapy Sharifi-Rad J, Quispe C, Patra JK , Singh YD , Panda MK , Das G, Adetunji CO , et al . Oxidative Medicine and Cellular Longevity.2021;2021. CrossRef
- Paclitaxel's Mechanistic and Clinical Effects on Breast Cancer Abu Samaan TM , Samec M, Liskova A, Kubatka P, Büsselberg D. Biomolecules.2019;9(12). CrossRef
- Characterization of acquired paclitaxel resistance of breast cancer cells and involvement of ABC transporters Němcová-Fürstová V, Kopperová D, Balušíková K, Ehrlichová M, Brynychová V, Václavíková R, Daniel P, Souček P, Kovář J. Toxicology and Applied Pharmacology.2016;310. CrossRef
- Cytochrome P450 3A4, 3A5, and 2C8 expression in breast, prostate, lung, endometrial, and ovarian tumors: relevance for resistance to taxanes Eijk M, Boosman RJ , Schinkel AH , Huitema ADR , Beijnen JH . Cancer Chemotherapy and Pharmacology.2019;84(3). CrossRef
- Resistance to Intervention: Paclitaxel in Breast Cancer Dan VM , Raveendran RS , Baby S. Mini Reviews in Medicinal Chemistry.2021;21(10). CrossRef
- Study of the Genetic Polymorphisms of ABCB1 3435G>A in Postmenopausal Women Breast Cancer on Paclitaxel Chemotherapy Hasan DAM , Sahib AS , Al-Rokan AAH , Mohsin KK . Journal of Contemporary Medical Sciences.2023;9(3). CrossRef
- Chemotherapeutic properties and side-effects associated with the clinical practice of terpene alkaloids: paclitaxel, docetaxel, and cabazitaxel Sousa-Pimenta M, Estevinho LM , Szopa A, Basit M, Khan K, Armaghan M, Ibrayeva M, et al . Frontiers in Pharmacology.2023;14. CrossRef
- Comparative effectiveness of early-line nab-paclitaxel vs. paclitaxel in patients with metastatic breast cancer: a US community-based real-world analysis Mahtani RL , Parisi M, Glück S, Ni Q, Park S, Pelletier C, Faria C, Braiteh F. Cancer Management and Research.2018;10. CrossRef
- Cancer-type organic anion transporting polypeptide 1B3 (Ct-OATP1B3) is localized in lysosomes and mediates resistance against kinase inhibitors Haberkorn B, Oswald S, Kehl N, Gessner A, Taudte RV , Dobert JP , Zunke F, Fromm MF , König J. Molecular Pharmacology.2022;102(6). CrossRef
- The involvement of human organic anion transporting polypeptides (OATPs) in drug-herb/food interactions Ali Y, Shams T, Wang K, Cheng Z, Li Y, Shu W, Bao X, et al . Chinese Medicine.2020;15. CrossRef
- Effects of SLCO1B1 and SLCO1B3 Genetic Polymorphisms on Valsartan Pharmacokinetics in Healthy Korean Volunteers Song G, Chung J, Yee J, Lee K, Park K, Gwak H. Journal of Personalized Medicine.2021;11(9). CrossRef
- Impact of SLCO1B3 polymorphisms on clinical outcomes in lung allograft recipients receiving mycophenolic acid Tague LK , Byers DE , Hachem R, Kreisel D, Krupnick AS , Kulkarni HS , Chen C, et al . The Pharmacogenomics Journal.2020;20(1). CrossRef
- Patients' Preference for Pharmaceutical Dosage Forms: Does It Affect Medication Adherence? A Cross-Sectional Study in Community Pharmacies Limenh LW , Tessema TA , Simegn W, Ayenew W, Bayleyegn ZW , Sendekie AK , Chanie GS , et al . Patient Preference and Adherence.2024;18. CrossRef
- Development of an automated chemiluminescent immunoassay for cancer antigen 72-4 and the evaluation of its analytical performance Yanagihara F, Okura H, Ichikawa H, Shirakawa T, Pan Y, Tu B, Lin Z, et al . Practical Laboratory Medicine.2023;34. CrossRef
- Current management of chemotherapy-induced neutropenia in adults: key points and new challenges: Committee of Neoplastic Supportive-Care (CONS), China Anti-Cancer Association Committee of Clinical Chemotherapy, China Anti-Cancer Association Ba Y, Shi Y, Jiang W, Feng J, Cheng Y, Xiao L, Zhang Q, et al . Cancer Biology & Medicine.2020;17(4). CrossRef
- Management of oral mucositis in patients who have cancer Lalla RV , Sonis ST , Peterson DE . Dental Clinics of North America.2008;52(1). CrossRef
- Chemotherapy-induced peripheral neuropathy Zhang X, Chen W, Huang W. Biomedical Reports.2017;6(3). CrossRef
- Allele-specific PCR in SNP genotyping Gaudet M, Fara A, Beritognolo I, Sabatti M. Methods in Molecular Biology (Clifton, N.J.).2009;578. CrossRef
- A review on breast cancer in Iraq and future therapies insights Alrawi N. Baghdad Journal of Biochemistry and Applied Biological Sciences.2022;3(01). CrossRef
- Adjuvant and Neoadjuvant Treatment of Triple-Negative Breast Cancer With Chemotherapy Marra A, Curigliano G. Cancer Journal (Sudbury, Mass.).2021;27(1). CrossRef
- A novel taxane, difluorovinyl-ortataxel, effectively overcomes paclitaxel-resistance in breast cancer cells Rong D, Wang C, Zhang X, Wei Y, Zhang M, Liu D, Farhan H, et al . Cancer Letters.2020;491. CrossRef
- The role of toll-like receptor 4 in tumor microenvironment Li J, Yang F, Wei F, Ren X. Oncotarget.2017;8(39). CrossRef
- Cytoskeletal Proteins in Cancer and Intracellular Stress: A Therapeutic Perspective Ong MS , Deng S, Halim CE , Cai W, Tan TZ , Huang RY , Sethi G, et al . Cancers.2020;12(1). CrossRef
- Influence of human OATP1B1, OATP1B3, and OATP1A2 on the pharmacokinetics of methotrexate and paclitaxel in humanized transgenic mice Steeg E, Esch A, Wagenaar E, Kenworthy KE , Schinkel AH . Clinical Cancer Research: An Official Journal of the American Association for Cancer Research.2013;19(4). CrossRef
- Variants in the SLCO1B3 gene: interethnic distribution and association with paclitaxel pharmacokinetics Smith N. F., Marsh S., Scott-Horton T. J., Hamada A., Mielke S., Mross K., Figg W. D., et al . Clinical Pharmacology and Therapeutics.2007;81(1). CrossRef
- Polymorphisms and linkage disequilibrium of the OATP8 (OATP1B3) gene in Japanese subjects Tsujimoto M, Hirata S, Dan Y, Ohtani H, Sawada Y. Drug Metabolism and Pharmacokinetics.2006;21(2). CrossRef
- Organic anion transporting polypeptides: Pharmacology, toxicology, structure, and transport mechanisms Hagenbuch B, Stieger B, Locher KP . Pharmacological Reviews.2025;77(2). CrossRef
- Elevated levels of serum tumor markers CA 15-3 and CEA are prognostic factors for diagnosis of metastatic breast cancers Lee JS , Park S, Park JM , Cho JH , Kim SI , Park B. Breast Cancer Research and Treatment.2013;141(3). CrossRef
- Pharmacogenetics of taxanes: impact of gene polymorphisms of drug transporters on pharmacokinetics and toxicity Jabir RS , Naidu R, Annuar MABA , Ho GF , Munisamy M, Stanslas J. Pharmacogenomics.2012;13(16). CrossRef
- Influence of polymorphic OATP1B-type carriers on the disposition of docetaxel Graan AM , Lancaster CS , Obaidat A, Hagenbuch B, Elens L, Friberg LE , Bruijn P, et al . Clinical Cancer Research: An Official Journal of the American Association for Cancer Research.2012;18(16). CrossRef
- Polymorphisms in cytochromes P450 2C8 and 3A5 are associated with paclitaxel neurotoxicity Leskelä S., Jara C., Leandro-García L. J., Martínez A., García-Donas J., Hernando S., Hurtado A., et al . The Pharmacogenomics Journal.2011;11(2). CrossRef
- Chemotherapy-induced peripheral neuropathy: Current status and progress Brewer JR , Morrison G, Dolan ME , Fleming GF . Gynecologic Oncology.2016;140(1). CrossRef
- Chemotherapy and its Adverse Effects – A Systematic Review Kaur S, Mayanglambam P, Bajwan D, Thakur N. International Journal of Nursing Education and Research.2022;10(4). CrossRef
- Evaluation of chemotherapy-induced cutaneous side effects in cancer patients Saadet ED , Tek I. International Journal of Dermatology.2022;61(12). CrossRef
- Association of genetic polymorphisms in SLCO1B3 and ABCC2 with docetaxel-induced leukopenia Kiyotani K, Mushiroda T, Kubo M, Zembutsu H, Sugiyama Y, Nakamura Y. Cancer Science.2008;99(5). CrossRef
- Polymorphisms in SLCO1B3 and NR1I2 as genetic determinants of hematotoxicity of carboplatin and paclitaxel combination Mbatchi LC , Schmitt A, Thomas F, Cazaubon Y, Robert J, Lumbroso S, Brouillet J, et al . Pharmacogenomics.2015;16(13). CrossRef
License

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
Copyright
© Asian Pacific Journal of Cancer Biology , 2026
Author Details