Checkpoint Inhibition in Microenvironment of Endometrial Cancer: The Emerging Role of KIR and NKG2A
Download
Abstract
The most common cancer of the female genital tract in developed nations is endometrial carcinoma (EC). Due in major part to population aging and the rising prevalence of diabetes and obesity, its incidence has grown by more than 132% globally over the last three decades. EC-related mortality has continued to rise even while improvements in early detection and treatment have raised survival rates. In contrast to other cancers like breast cancer, less is known about the immunological landscape of EC. Although T cell-based immunotherapies have revolutionized cancer treatment, certain tumor types and patient populations show limited responsiveness. As a result, the innate immune system in particular, natural killer (NK) cells has drawn more attention as a potential substitute effector in cancer immunotherapy. NK cells are powerful antitumor cytotoxic lymphocytes that are frequently compromised in the tumor microenvironment. These cells are controlled by a balance of activating and inhibitory receptors that recognize the lack of MHC class I molecules. Current treatment approaches block inhibitory receptors or stimulate activating ones in an effort to improve NK cells-mediated anticancer responses. With an emphasis on inhibitory receptors, including killer cell immunoglobulin-like receptors KIR and NKG2A, this review investigates the phenotypic and functional features of NK cells within the TME of EC. Understanding how modulation of KIR and NKG2A signaling can restore NK cells cytotoxicity provides a promising avenue for next-generation immunotherapies in EC.
1. Introduction
With around 10 million deaths in 2022, cancer continues to rank as the second most common cause of death globally. By 2040, it is expected to be about 28 million new cases of cancer worldwide and about 16 million deaths from this disease. Gynecological malignancies, which mostly affect the female reproductive organs the endometrium, cervix, ovaries, and vulva represent a major public health concern among the several forms of cancer [1]. Endometrial carcinoma (EC) is the most prevalent gynecologic cancer in developed nations, and it originates from the uterine lining [2]. EC was the sixth most common cancer diagnosed in women globally in 2020 [3]. Although postmenopausal women are the majority of those affected by EC, its occurrence has been rising rapidly among younger women, primarily as a result of risk factors such as hormonal imbalances like polycystic ovarian syndrome (PCOS) and obesity [2]. The global incidence of EC has risen significantly over the past three decades. Approximately 417,000 new cases and 97,370 deaths were reported in 2020 alone (Table 1) [4].
| Cancer Type | Worldwide New Cases | Worldwide Deaths | Worldwide Female Cancer Rank | 5-Year Survival Rates (United States) |
| Cervical Cancers | 604,127 | 341,831 | 5 th | ~66% in the whole population; Low stage: 92% and 58% if lymph nodes are invaded |
| Endometrial Cancers | 417,367 | 97,370 | 7 th | Early stages: >80% (95% for stage I); Recurrent/advanced disease: 20-25% |
| Ovarian Cancers | 313,959 | 207,252 | 9 th | ~49.7% |
| Vaginal Cancers | 17,908 | 7,995 | >10 th | ~49% in the whole population with variation 35–78%; Early stage: ~85% |
| Vulvar Cancers | 45,240 | 17,427 | >10 th | ~70.3% |
The main causes of this trend are the aging of the population and an increase in obesity and diabetes [3].
Age-standardized mortality rates have decreased in many areas as a result of developments in early detection and treatment, even though the number of deaths from EC has nearly doubled since 1990 [6]. With a positive prognosis and five-year survival rates above 80%, the majority of EC patients are identified early (Figure 1).
Figure 1. Cancer Stat Facts: Uterine Cancer [8].
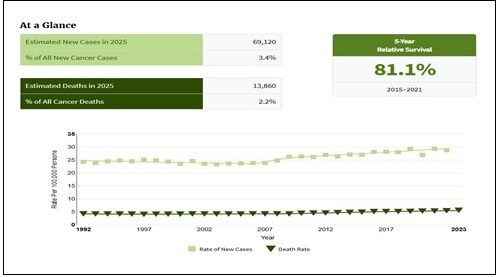
However, extrauterine illness affects up to 20% of patients at presentation, and in stage IV cases, the five-year survival rate reduces to 15% [7].
Histologically, EC has been divided into two primary categories based on Bokhman’s dualistic paradigm, which was first presented in 1983. Estrogen-dependent type I EC frequently develops in the context of endometrial hyperplasia and unopposed estrogen exposure. These types of cancer have a good prognosis, typically appear endometrioid in histology, are low-grade, and exhibit less aggressive clinical behavior. On the other hand, Type II EC is not dependent on estrogen and comprises more aggressive histological subtypes such as carcinosarcoma, clear cell carcinoma, and serous carcinoma. These tumors are frequently of high grade and are associated with worse clinical results [9]. The International Federation of Gynecology and Obstetrics (FIGO) estimates that Type I tumors, which include grades 1 and 2, account for approximately 80% of EC cases, whereas Type II tumors, which include grade 3, comprise roughly 10% to 20% [10]. However, despite Bokhman’s classification’s historical significance and widespread application, this binary model has shown low consistency and clinical accuracy, mostly because the two categories’ features overlap, and it is unable to adequately reflect the molecular heterogeneity of EC [11]. The Cancer Genome Atlas (TCGA) introduced a molecular classification system in 2013 to overcome these limitations while greatly enhancing prognostic stratification. The system identifies four molecular subgroups: copy-number low, copy-number high, microsatellite instability-high (MSI-H/hypermutated), and POLE-ultramutated. The MSI-H subgroup, characterized by a defect in mismatch repair (dMMR), exhibits a strong response to immune checkpoint blockade (ICB) therapies [12]. The National Comprehensive Cancer Network (NCCN) recommendations (2021) suggested universal molecular profiling for all EC diagnoses, given its clinical significance. Additionally, the ESGO/ESTRO/ESP guidelines have incorporated these molecular subtypes into risk stratification algorithms to guide adjuvant therapy decisions [13]. Importantly, these molecular groups are also associated with distinct tumor immune microenvironments, influencing immune cell infiltration and responsiveness to immunotherapy [14].
EC is often treated by a total hysterectomy, bilateral salpingo-oophorectomy, and pelvic lymph node dissection. Adjuvant chemotherapy or radiation therapy is frequently used after these procedures [15]. These treatments are frequently effective for EC in its early stages, but there are still few treatment choices available for severe or recurring illness. Although platinum–taxane chemotherapy is usually used as the first line of treatment, there isn’t yet a second-line standard that is widely recognized. Furthermore, especially in cases of platinum-resistant illness, individuals with comorbidities or decreased performance status may not tolerate systemic chemotherapy well. These difficulties emphasize how urgently novel and focused therapeutic strategies, like immunotherapy, are needed [5]. EC is becoming more commonly recognized as an immunologically regulated cancer in which metastasis, treatment resistance, and disease progression are all significantly influenced by the immunosuppressive tumor microenvironment (TME) [16]. The TME in EC is composed of a diverse group of immunological infiltrates that may exhibit both pro- tumor and anti-tumor effects, including T cells, B cells, macrophages, dendritic cells, and natural killer (NK) cells [2]. While the effectiveness of immune checkpoint blockage and CD8 T cell exhaustion have been well studied [17], the function of NK cells in EC is still mostly unknown.
NK cells are cytotoxic innate lymphoid cells essential for the early stages of anti-tumor immunity. Directly destroying tumor cells and secreting cytokines are two of their effector actions. NK cells can target tumor cells with MHC-I downregulation, a frequent immune evasion strategy, because they do not require MHC-I– mediated antigen presentation like CD8+ T cells do. However, NK cells activity is often suppressed inside the immunosuppressive TME [18]. A delicate balance between activating and inhibiting receptors controls the function of NK cells. NK cells dysfunction can be triggered by suppressive cytokines like TGF-β, IL-6, and IL-32a as well as inhibitory receptors such as killer cell immunoglobulin-like receptors (KIRs), NKG2A, immunoglobulin-like transcript receptors (ILTs), and TIGIT. On the other hand, NK cells cytotoxicity is promoted by activating receptors, such as DNAM-1, NKp80, KIRs, NKG2D, and natural cytotoxicity receptors (NCRs), as well as cytokines, such as IL-12, IL-15, and IL-18. NK cells are powerful during the elimination stage of carcinogenesis, but as tumors grow, they frequently lose their ability to function [19]. NK cells specifically the CD56bright CD16dim subset represent an achievable focus in the treatment of EC due to their unique functional properties and potential as immunotherapy targets. With a focus on the functions of inhibitory receptors, including killer cell immunoglobulin-like receptors (KIRs), NKG2A, and their ligands HLA-C and HLA-E, this review aims to investigate the regulatory mechanisms affecting NK cells function in EC. The development of novel immunotherapeutic approaches that reactivate or strengthen NK cells-mediated anti-tumor responses may be made possible by a thorough understanding of the processes behind these checkpoints, which would eventually improve clinical outcomes for EC patients.
2. Molecular Classification of Endometrial Cancer
A molecular-based classification system was introduced by The Cancer Genome Atlas (TCGA) in 2013, and it completely changed the prognosis and treatment landscape for EC (Figure 2).
Figure 2. Molecular Subtypes of EC [16].
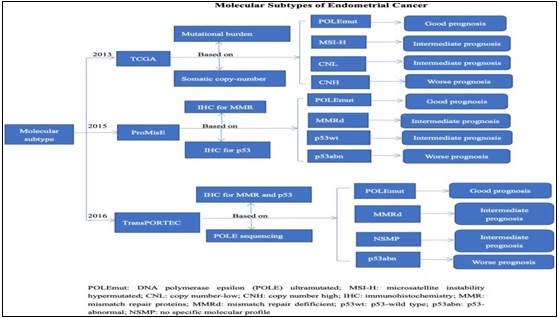
Four different molecular subtypes were found using this system: POLE-ultramutated: a condition with an extraordinarily high rate of mutation and a favorable prognosis; microsatellite instability-high (MSI-H): linked to a positive reaction to immune checkpoint drugs and high immunogenicity; copy-number low (CNL) indicates a prognosis that is in between, and poor prognosis is linked to copy-number high (CNH)/p53-abnormal, which usually comprises serous-like tumors. Beyond its prognostic value, this classification reflects distinct tumor immune microenvironments (TME), which are particularly relevant in the MSI-H and POLE-mutated subtypes, where responsiveness to immunotherapy is notably enhanced [12].
2.1 Clinical Translation of TCGA: ProMisE and TransPORTEC
ProMisE (Proactive Molecular Risk Classifier for Endometrial Cancer) is a surrogate diagnostic platform designed to make it easier to apply TCGA’s molecular subtypes in clinical settings. Talhouk et al., developed the ProMisE for Endometrial Cancer (ProMisE), which utilizes immunohistochemistry to identify mismatch repair (MMR) proteins, including mutL homolog 1 [20], PMS1 homolog 2, mismatch repair system component, mutS homolog 2, and mutS homolog 6, as well as p53 expression and DNA sequencing to identify POLE mutations. The feasibility of the ProMisE system has recently been validated in 452 EC cases. These systems offer a feasible and cost-effective approach for molecular classification in clinical settings. The clinical implications of these molecular subtypes are now integrated into treatment guidelines. In 2016, Stelloo et al., validated a more pragmatic, cost-effective, and clinically applicable molecular classification system called the Translational Research in Post-Operative Radiation Therapy in EC (TransPORTEC) system. Therefore, EC can be subdivided into four subgroups: the POLE-mutated, MMRd, p53abn, and no specific molecular profile (NSMP) [21, 22]. POLE-mutant ECs have a highly favorable prognosis and do not require adjuvant treatment. MMRd ECs have an intermediate prognosis. Numerous recent studies have focused on immunotherapy for EC [23, 24]. Furthermore, these two groups may benefit from immunotherapy. P53-mutant ECs are typically associated with more advanced stages, higher rates of lymphatic vascular space infiltration (LVSI), and diverse pathological types. Most P53mutant ECs are serous adenocarcinomas and have a poor prognosis. The NSMP group was the most frequent, and its prognosis was uncertain [22], as shown in (Figure 2). Recent consensus guidelines by ESGO/ ESTRO/ESP have integrated molecular classification into risk stratification models, enabling personalized treatment decisions based on underlying tumor biology. This framework also enhances understanding of immune- related mechanisms such as natural killer (NK) cell infiltration, dysfunction, and immune escape which are known to vary significantly among the molecular subtypes [13]. IHC-based investigations have been performed to further investigate the relation between tumor-infiltrating immune cell characteristics and molecular classification. The quantity and immunological characteristics of invading immune cells, especially in POLEmut and MMRd subtypes, have been confirmed by numerous investigations. These characteristics are thought to play a role in the better survival rates and increased immunotherapy response shown in these populations [25].
3. Immune Microenvironment of the Normal Endometrium: Foundation for Understanding EC Immunity
Examining the endometrium’s normal immunological milieu is necessary before comprehending the immune features of EC. The uterine microbiota, pathogen exposure, and hormone oscillations all contribute to the endometrium’s cyclical changes in immune cell composition and activity. These immune cells enable tissue remodeling during menstruation, implantation, and pregnancy, in addition to defending the endometrium against infections. During the roughly 400 menstrual cycles between menarche and menopause, the endometrium, a hormonally controlled mucosal tissue, has repeated tissue breakdown and regeneration that is predominantly driven by estrogen and progesterone [26]. These reproductive hormones often create an immunosuppressive environment that promotes conception and are thought to be the main regulators of endometrial immune activity [15]. In particular, during the secretory phase, progesterone and estrogen influence immune cell recruitment, activation, and activity, fostering an immunologically tolerant state. Although necessary for optimal reproduction, this hormone-driven immunosuppression also reflects several features of immune evasion observed in the EC tumor microenvironment. Both innate and adaptive immune cells make up the endometrial immunological landscape (Figure 3).
Figure 3. Immune Cell Population Disruption in Normal Endometrial Tissue [15].
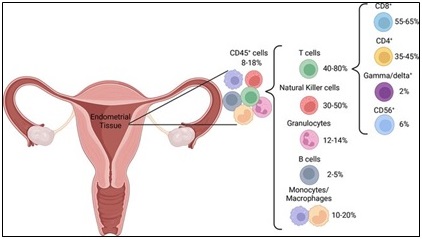
T cells, dendritic cells, and regulatory T cells make up a smaller but crucial population, but innate lymphoid cells mainly natural killer (NK) cells and macrophages are the majority. B cells are rare and mostly localized within lymphoid aggregates (LAs), which cyclically form during the late proliferative phase, enlarge in the secretory phase, and disappear after menopause. Immune cell composition fluctuates across the menstrual cycle, with T cells dominating the proliferative phase and NK cells increasing significantly during the secretory phase. After menopause, T cell levels stabilize, while granulocytes become more abundant due to tissue atrophy [15]. Among the immune cells present in the endometrium, uterine natural killer (uNK) cells are especially sensitive to hormonal cues and interleukin-15 (IL-15). Progesterone, in particular, enhances their migratory and functional capabilities. uNK cells begin to appear early in the menstrual cycle, proliferate during the secretory phase, and are ultimately cleared by apoptosis and shedding if fertilization does not occur [27]. During the secretory phase and early pregnancy, uterine NK (uNK) cells particularly the CD56^bright CD16^dim subset play a key role in decidualization. They facilitate vascular remodeling, trophoblast invasion, and placental development while maintaining immune tolerance. Their activity is modulated by reproductive hormones, especially estrogen and possibly luteinizing hormone (LH), which may enhance their adhesion and accumulation in the endometrial tissue. These cells favor cytokine production over cytotoxicity unless exposed to pathogens [15, 27]. Macrophages constitute a significant portion of the CD45+ immune population in the endometrium. More than 70% co-express CD68 and CD163, indicating an M2 anti-inflammatory phenotype. These macrophages contribute to tissue repair, angiogenesis, and immune regulation, secreting cytokines like IL-10. Their polarization supports an immunosuppressive environment favorable for embryo implantation and pregnancy maintenance [15].
The endometrial immunological environment is greatly impacted by systemic diseases like obesity in addition to hormonal factors. Obesity is a significant risk factor for EC due to immunological regulation as well as hormonal mechanisms, such as increased adipose tissue synthesis of estrogen. By decreasing CD8 T cell infiltration and NK cell’s function, it creates a suppressive tumor microenvironment that may accelerate tumor growth and reduce immune surveillance. Remarkably, it has been demonstrated that immune cell activity in the endometrium is restored by weight loss. Ironically, certain EC patients may respond better to immune checkpoint inhibitors if their BMI is higher, indicating a complex relationship between obesity and the effectiveness of immunotherapy [15]. Carcinogenesis, which includes EC, has been linked to the dysregulation of the immune system, particularly persistent unresolved inflammation. In patients with EC, elevated systemic markers, including C-reactive protein (CRP), have been suggested as prognostic indicators [27, 28]. Understanding how immune dysregulation, influenced by both internal and external stimuli, plays a role in the etiology and development of EC is made easier by these immunological characteristics of the healthy endometrium.
4. Endometrial Cancer’s Immune Microenvironment and Immune Escape Mechanisms
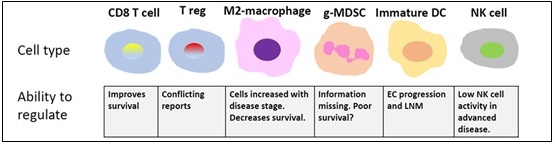
A complex network of immune cell populations, stromal cells, cytokines, chemokines, and signaling molecules makes up the immunological microenvironment in EC, which together have both tumor-promoting and protective effects (Figure 4).
Figure 4. The Microenvironment of EC Tumors Contains Primary Immune modulatory Cells. There are several different kinds of cells in the EC tumor microenvironment. Many of which have low survival rates and favorable progression [10].
By identifying and destroying cancerous cells, immune effector cells, including natural killer (NK) cells and cytotoxic T lymphocytes (CTLs), act as front-line defenses. Tumor-derived signals, on the other hand, can change this microenvironment and create an immunosuppressive tumor microenvironment (TME), which promotes immune evasion and tumor growth [2]. As previously mentioned, the TME plays a role in angiogenesis, tissue remodeling, and immunomodulation,among other tumor-supporting mechanisms. While cytokines and chemokines promote cancer invasion and growth and influence immune cell recruitment toward suppressive phenotypes, stromal components especially cancer-associated fibroblasts (CAFs) and extracellular matrix components offer structural and regulatory support [2]. To avoid immune surveillance, EC cells use a variety of immune evasion techniques (Figure 5).
Figure 5. Tumor-infiltrating Immune Cells and EC Interact Intricately to form a Complex Microenvironment where Conflicting Forces Can Either Promote or Inhibit Tumor Growth. The dynamic equilibrium that immune cells preserve inside the tumor microenvironment is the source of this dichotomy. A cadre of cells that specialize in immune evasion promotes tumor growth, even though some immune components have anti-tumor actions. This intricate immune system emphasizes the necessity of comprehending the intricate molecular processes involved to better apply this understanding to tailored approaches and efficient therapies [2].
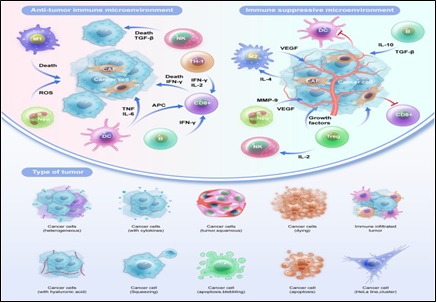
One important mechanism is the overexpression of immunological checkpoint molecules, such as programmed death-ligand 1 (PD-L1), which attach to T cell PD-1 receptors to cause T cell fatigue and prevent cytotoxic activity. Furthermore, EC cells release immunosuppressive cytokines that further inhibit T cell activation and NK cells cytotoxicity, such as transforming growth factor-beta (TGF-β) and interleukin-10 (IL-10). Tumor cells are less visible to immune effectors when tumor-associated antigens (TAAs) and major histocompatibility complex (MHC) molecules are downregulated [29, 30].
The EC microenvironment is frequently enriched in immunosuppressive cell types, including regulatory T cells (Tregs) and myeloid-derived suppressor cells (MDSCs). By reducing effector T cell proliferation and cytokine generation, these cells actively decrease anti-tumor immune responses, fostering tolerance toward the tumor. The physiological immunological tolerance of the normal endometrium, which is necessary for embryo implantation and pregnancy maintenance, is maladaptively extended in this pathological immunosuppressive state. This balance is disrupted in EC, leading to a microenvironment that promotes tumor development, invasion, and metastasis by means of coordinated interactions between soluble mediators, stromal elements, and immune cells (Figure 6) [2].
Figure 6. Programmed Death-1 (PD-1)/programmed death-ligand 1 (PD-L1) Axis is one of the Immunological Checkpoint Mechanisms Utilized Strategically by EC Cells. As demonstrated, the complex process entails cancer cells upregulating PD-L1 expression to enable them to communicate with T cells' PD-1. The anti-tumor immune response is effectively suppressed by this interaction, which causes T cell fatigue and anergy. Numerous studies have highlighted the importance of these pathways in gynecological malignancies and acknowledged their therapeutic relevance. Deciphering these intricate relationships yields vital information for creating immunotherapies that are specifically designed to effectively combat EC [2].
Ultimately, one of the main obstacles to successful immunotherapy is the immunosuppressive character of the EC tumor microenvironment (Table 2).
| Cell type | Regulation | Specimen | Clinical application |
| CD8+ T cells | Down | Blood and tumor tissue | Presence at the invasive border predicts survival in EC. |
| Treg cells | Up | Blood and tumor tissue | Associated with tumor grade, stage, and myometrial invasion. CD4+CD25+ and CD4+Foxp3+/CD8+ ratios are prognostic. |
| Macrophages | Up | Tumor tissue | High CD68+ density correlates with worse PFS/OS, higher grade, LVI, LNM; predicts recurrence-free survival and OS in type-I EC. |
| NK cells | Down | Blood and tumor tissue | Reduced NK activity linked to myometrial invasion; NK cells activity and HLA-E expression associate with survival/prognosis. |
| Dendritic cells | Down | Tumor tissue | S100 and HLA-DR markers delay tumor progression and LNM in EC. |
| B lymphocytes | Up | Tumor tissue | Low T cell/NK/macrophage markers; increased CD20 and S100 expression associated with poor outcomes. |
Therefore, the development of treatment options aiming at reactivating immune surveillance and enhancing clinical outcomes requires a thorough understanding of the cellular and molecular components that maintain immune evasion [2].
4.1 T Cells and Regulatory T Cells (Tregs) in Endometrial Cancer
T cells, especially the CD4+ (helper) and CD8+ (cytotoxic) subsets, are essential for immunological infiltration in EC [18]. CD4+ T helper cells organize the immune response by triggering other immune cells and releasing cytokines, whereas CD8+ cytotoxic T lymphocytes (CTLs) promote tumor cell death by apoptosis. Nevertheless, little is known about the function of T cells in the tumor microenvironment (TME) in EC [2]. According to several studies, EC tissues have fewer CD8 T lymphocytes than non-malignant endometrium. For instance, EC patients had significantly reduced CD8+ T cell numbers and expression, according to a study comparing malignant and non-malignant samples; in contrast, low-grade tumors with more CD8+ infiltration at invasive tumor borders had longer overall survival (OS) [31]. Pascual-García et al., and Kondratiev et al., identified intraepithelial CD8+ T cell density as an independent predictor of OS alongside stage, vascular invasion, and tumor grade [31, 32]. Suemori et al., and Dai et al., found that higher CD8+ CTL levels correlate with improved prognosis, especially in tumors with high tumor mutational burden (TMB) producing more neoantigens, while tumors with low TMB and late-stage disease show reduced CD8+ infiltration [33, 14].
The EC molecular subtype modulates CD8 T cell density and activity. Tumor-infiltrating lymphocytes (TILs), including granzyme B+ and PD-L1+ CD8+ T cells, are abundant in MSI-H and POLE-mutated tumors and are associated with better prognosis. While Kim et al., found that a high PD-L1+ T cell infiltration was associated with a shorter progression-free survival (PFS) in microsatellite-stable (MSS) cancers [34]. Willvonseder et al., found that high-grade ECs had higher TILs and PD-1/PD-L1 expression [35]. These findings demonstrate that the mutational and immune landscape of EC determines the functional outcome of CD8⁺ infiltration, with immunogenic subtypes (MSI-H and POLE-mutated) showing more active cytotoxic responses and better clinical outcomes.
Conversely, regulatory T cells (Tregs), primarily CD4+CD25+FoxP3+ subsets that inhibit anti-tumor immunity, infiltrate EC more frequently. Chang et al., found that tumor-infiltrating lymphocytes (TILs) had substantially greater CD4+CD25+ Treg counts than peripheral blood, which were correlated with the myometrial invasion depth, tumor grade, and stage [36]. According to Yamagami et al., prognostic indicators include CD4FoxP3FoxP3 cell counts and CD4FoxP3FoxP3/CD8FoxP3 ratios [37]. Tregs can limit effective immune responses by reducing NK and CD8 T cells within the TME by expressing granzyme B and perforin [2]. Zhang et al., and Julie et al., also reported that a high Treg/CD8⁺ CTL ratio correlates with aggressive features, such as lymph node metastases and vascular invasion [16, 38]. Furthermore, CD8+ TILs in EC frequently exhibit decreased expression of granzyme B and perforin, which reduces their capacity to cause cytotoxicity [11]. Antigen-presenting cells (APCs), which stimulate CD4+ T cells and encourage CD8+ and NK cells cytotoxicity via Th1 polarization, also have an impact on adaptive immunity in EC. However, by altering the Treg/ Th1 ratio, tumor-derived substances may inhibit APC function, shifting the immunological balance in favor of immunosuppression. Potential therapeutic targets, this immune modulation leads to immunological escape in EC and matches changes observed during pregnancy (Th1/ Th2 and Treg/Th17) [11].
4.2 B Lymphocytes in EC Tumor Microenvironment
In the tumor microenvironment (TME) of EC, B lymphocytes are less abundant than T cells. However, they are nevertheless important in regulating immune responses. Numerous investigations have located B cells in EC tissues and emphasized their diverse roles in tumor immunity. Zinovkin and Pranjol showed that downregulation of lineage-specific markers in T lymphocytes (CD3), natural killer (NK) cells (CD57), and macrophages (CD68), in conjunction with upregulation of B lymphocyte (CD20) and dendritic cell (S100) markers, was linked to worse clinical outcomes in a study of 82 patients with endometrioid adenocarcinoma [39]. These results highlight the intricate interactions among immune cell types that shape the TME. By producing cytokines and antibodies, B cells support anti-tumor immunity. They can increase the death of tumor cells by producing antibodies that target tumor-associated antigens through a process known as antibody-dependent cell-mediated cytotoxicity (ADCC). Furthermore, the function of B cell-rich tertiary lymphoid structures (TLSs) in maintaining local immunological responses is supported by their presence inside the TME [15]. Remarkably, better survival rates for EC patients are correlated with T and B cell co-infiltration. Regulatory B cells (Bregs), specifically the CD19+CD24^hiCD27+ subgroup, secrete interleukin-10 (IL-10), which has immunosuppressive properties. IL-10 controls Th1 type inflammation, prevents T cell activation, and keeps dendritic cells immature. Interestingly, lower levels of Bregs have been observed in women who miscarried in the early stages of pregnancy, indicating that immunological tolerance mechanisms are similar throughout pregnancy and during the growth of tumors [40]. Despite their very limited infiltration in EC, new research indicates that B cells might have a major impact through processes that are not dependent on antigen. For instance, dimeric IgA, but not monomeric IgG, binds to the polymeric immunoglobulin receptor (pIgR) and triggers pro-apoptotic, endoplasmic reticulum (ER) stress, and intracellular inflammatory pathways in tumor cells, all of which enhance patient outcomes. It is noteworthy that pIgR is nearly always expressed in EC cells, suggesting that this pathway may be important for EC immunobiology. Furthermore, TLSs continue to be a significant source of B cells inside the tumor, highlighting their functional and geographical importance in the TME [15].
4.3 Tumor-associated Macrophages (TAMs)
A key component of the tumor microenvironment (TME) in EC, tumor-associated macrophages (TAMs) promote invasion and angiogenesis, which promotes tumor growth, metastasis, and therapy resistance [16]. Derived from circulating monocytes, TAMs retain common cytotoxic and phagocytic functions but are often polarized into two phenotypes: M1 macrophages, which are pro-inflammatory and anti-tumorigenic, expressing MHC I/II and producing IL-12, IL-23, and TNF-α; and M2 macrophages, which are immunosuppressive and pro-tumorigenic, secreting IL-10 and TGF-β [15, 16]. TAMs in EC mostly take on the M2 phenotype, which promotes an aggressive tumor behavior and immunosuppressive TME [11, 15].
Among the most prevalent immune cells in EC are TAMs, which frequently outnumber CD8+ cytotoxic T lymphocytes [10, 16]. Their predictive significance is still up for discussion, though. Other studies have connected high TAM levels to poor clinical outcomes, but Van Der Woude et al. could not find a strong linkage between TAM density and the course of cancer [15]. Kübler et al., linked shorter overall survival (OS), recurrence- free survival (RFS), and advanced illness to increased TAM expression [41]. Similarly, Soeda et al., associated a high intratumoral CD68+ TAM density with worse OS and PFS [42], whereas Dun et al., found enhanced CD68+ macrophages in both the epithelial and stromal compartments of type I and II EC [43]. Furthermore, Weber et al., found that advanced-stage EC had larger levels of CD163+ M2 macrophages [44], and Kübler et al., observed robust correlations with increased tumor grade, lymphovascular invasion, and lymph node metastasis [41]. Notably, M1/M2 polarization in EC parallels immune tolerance mechanisms in pregnancy, where M2 macrophages preserve fetal–maternal immune privilege via IL-10 secretion. Tumors exploit this mechanism to evade immune surveillance and promote disease progression [2, 40]. Therapeutic strategies targeting TAMs are under investigation, including inhibition of macrophage recruitment via CCL2/CCL5 blockade, depletion using agents like Trabectedin, and disruption of VEGF signaling with monoclonal antibodies such as Bevacizumab. These approaches aim to reprogram the TME towards a more immunostimulatory profile and enhance therapeutic efficacy [45].
4.4 Dendritic Cells (DC)
The most potent antigen-presenting cells (APCs) are dendritic cells (DCs), which are important in triggering adaptive immune responses by capturing, processing, and presenting tumor antigens to T lymphocytes. The ability of these cells to activate cytotoxic T lymphocytes (CTLs) is frequently compromised in EC because of the tumor microenvironment (TME), which results in reduced anti-tumor immunity and the advancement of the illness [10]. Common morphological and functional defects of tumor-infiltrating DCs in EC include the downregulation of co-stimulatory molecules such as CD80, CD86, and CD40 in comparison to normal endometrial tissue. This reduced expression hinders their ability to present antigens, restricts the activation of T cells, and facilitates immune evasion. It has also been associated with lymph node metastases, advanced clinical stage, and a poor prognosis [2, 10, 11, 16]. Moreover, immature DCs recruited to the tumor site can aggravate immune suppression, and an inverse relationship has been reported between activated DCs and CD8+ T cell density, where high DC infiltration correlates with poorer overall survival [16]. Jensen et al., for instance, found that plasmacytoid DCs (CD123+) within tumors were linked to unfavorable outcomes in EC patients [46]. Parallels have been drawn between the tolerogenic functions of DCs in pregnancy and in EC. In the decidua, CD83+ DCs maintain a Th2-dominant, immune-tolerant state via immunosuppressive cytokines a mechanism that appears hijacked by EC cells. Similarly, antigen-presenting markers like S100 and HLA-DR have been implicated in lymph node metastasis and adverse prognosis [47]. Additionally, reduced tumor antigenicity in EC hinders DC-mediated activation of CD8+ CTLs, further facilitating immune escape. Therapeutically, strategies aiming to restore DC function such as DC vaccines and cytokine modulation are under clinical evaluation. Although promising in other malignancies, DC-based therapies in EC are still in early stages and require more validation to improve patient outcomes [10, 16].
4.5 Myeloid-derived suppressor cells (MDSCs)
A diverse group of immature myeloid cells known as myeloid-derived suppressor cells (MDSCs) proliferate in the tumor microenvironment (TME) and are important in inhibiting anti-tumor immunity [48]. They suppress the immune system in a number of ways, such as by producing inhibitory cytokines like transforming growth factor-beta (TGF-β) and interleukin-10 (IL-10), as well as by preventing T cell proliferation through the action of arginase-1 and nitric oxide (NO) [2, 10, 27]. Notably, the enzyme inducible nitric oxide synthase (iNOS), which changes arginine into nitric oxide, is also expressed by MDSCs. Immune surveillance is further compromised by this nitric oxide generation, which hinders antibody-dependent cellular cytotoxicity (ADCC) mediated by natural killer (NK) cells (Figure 7) [27].
Figure 7. Dysregulation of NK Cells in Immunosuppressive UCIM. NK cells dysregulation and reduction are caused by elevated numbers of MDSCs and AAMs in the tumor microenvironment (TME), which also raise TGF-β1, NO, and arginase and lower IFN-γ levels. Dysregulation and depletion of NK cells can also be caused by the hypoxic environment found in some uterine cancers [27].
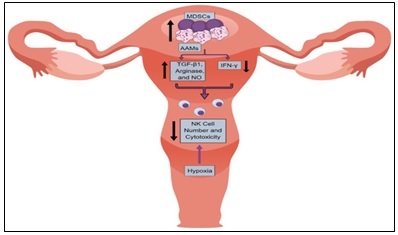
By interfering with both innate and adaptive immune responses, these pathways impair immune surveillance and encourage the growth of tumors. MDSCs aid in the formation of an immunosuppressive TME in EC, which promotes tumor angiogenesis, invasion, and immunotherapy resistance in addition to suppressing cytotoxic lymphocyte function. Their interactions with M2-polarized macrophages and regulatory T cells (Tregs), among other suppressive immune cells, illustrate the complex pathways of immune evasion in EC. MDSCs have become interesting therapeutic targets because of their crucial role. To improve the effectiveness of immune checkpoint inhibition and dendritic cell-based immunotherapies by reversing immune suppression and restoring potent anti-tumor responses, strategies to deplete MDSCs, reprogram their activity, or prevent their recruitment are being researched [2].
Collectively, the coordinated actions of immunosuppressive cells such as MDSCs, Tregs, and M2 macrophages not only suppress cytotoxic lymphocyte function but also promote the upregulation of immune checkpoint molecules and altered MHC expression, facilitating immune escape in EC.
4.6 Molecular Players of the Immune Response in EC: MHC Molecules and Immune Checkpoints
Major histocompatibility complex (MHC) molecules are essential for immune surveillance and antigen presentation. Both traditional and non-classical MHC molecules are expressed by epithelial cells in the normal endometrium, facilitating immune recognition. However, in EC, tumor cells often upregulate the non-canonical MHC class I molecule HLA-G and downregulate MHC class I and II molecules. This immune evasion strategy is similar to the immune privilege observed during pregnancy, when fetal trophoblasts express HLA-G to suppress maternal immunity. Inhibiting NK cells cytotoxicity, suppressing cytotoxic T lymphocyte (CTL) responses, modulating cytokine production, inducing immune cell death, and influencing stromal cell function are only a few of the immunosuppressive actions of HLA-G, which support tumor immune escape [40, 49].
Downregulating the conventional HLA class I molecules (HLA-A, -B, and -C) limits the presentation of antigens and allows malignancies to avoid being destroyed by CD8-CTLs. In 41.3% of sporadic endometrioid EC cases, de Jong et al., discovered deletion of HLA-A and/ or HLA-B/C, particularly in high-grade tumors [50]. Concurrently, Barrier et al., showed HLA-G protein expression in the glandular epithelium of many EC tumors [51]. All HLA-G isoforms (G1–G7) were present across different EC stages, though no clear correlation was found between their expression and survival. However, soluble isoforms particularly HLA-G5 were significantly elevated in EC patients, especially in early-stage disease, implying a role in shaping an immunosuppressive tumor microenvironment.
Immunological checkpoint circuits are essential immunological escape strategies in EC, in addition to MHC alteration. With PD-L1 expression present in 67– 100% of initial, recurrent, and metastatic EC lesions, the PD-1/PD-L1 axis is clearly engaged. Poorly differentiated tumors, non-endometrioid subtypes, and decreased progression-free survival (PFS) are linked to high PD-L1 expression, especially in situations where microsatellite instability (MSI) is present. Additionally, associated with poorer results are elevated PD-L1/CD8 and PD-L1/PD-1 ratios [11]. Furthermore, PD-L1 expression on tumor- associated T cells may act not only as a ligand but also as a receptor, triggering immunosuppressive signaling and inhibiting T cell differentiation into effective effector cells an added mechanism of immune evasion [16]. B7 family members B7-H3 and B7-H4 are examples of additional immune checkpoint molecules that aid in immunological escape. Strong B7-H3 expression was found in advanced EC by Brunner et al., and this expression was linked to both enhanced CD8+ TIL infiltration and poor overall survival (OS) [52]. Additionally, often overexpressed in malignant and hyperplastic endometrial tissues, B7- H4 is linked to aggressive tumor characteristics. B7-H4 expression appears to be unaffected by tumor grade, histological type, or immune infiltration, in contrast to other markers, indicating that it may be a universal immune escape marker in EC [52]. In conclusion, dysregulation of MHC molecule expression and upregulation of immune checkpoints are major contributors to immune evasion in EC. Immunotherapeutic treatments, especially immune checkpoint inhibitors, which seek to restore anti-tumor immunity and enhance clinical results, can effectively target these pathways.
5. Uterine Natural Killer (uNK) Cells
5.1 uNK Cells Phenotype, Disruption, and Development
Large granular lymphocytes of the innate immune system, natural killer (NK) cells may identify and destroy virus-infected or cancerous cells without requiring prior sensitization or MHC limitation. They are therefore interesting targets for immunotherapy and potent antitumor immune effectors [53]. Although some of these markers may overlap with specific T cell subsets, NK cells are characterized phenotypically by the expression of CD56 (NCAM) and CD16 (FcγRIIIA) and the lack of CD3 (Figure 8).
Figure 8. Different Expression Patterns for NK Cells subsets' Inhibitory Receptors. Different inhibitory receptors regulate terminally differentiated CD56dimCD57+ NK cells, mature CD56dim, and immature CD56bright NK cells. Red points to T cell-associated immune checkpoints that are up-regulated upon activation, green points to T cell-associated immune checkpoints expressed in steady state, blue points to MHC-I binding receptors, yellow points to receptors binding to nectin or nectin-like molecules, and purple points to other inhibitory pathways [55].
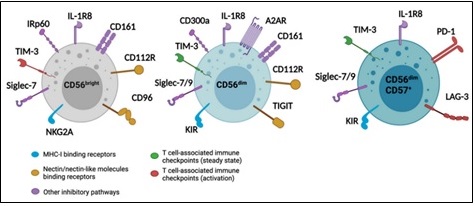
As a result, NK cells are usually identified by their CD3⁻ CD56+ phenotype [54]. They are separated functionally into two main subsets according to the expression of CD56: about 90% of peripheral blood NK cells are CD56dim CD16+ NK cells, characterized by strong cytotoxic activity and high expression levels of granzymes and perforin. Direct target cell death and antibody-dependent cellular cytotoxicity (ADCC) are their main functions [54], and the uterine decidua and secondary lymphoid tissues are rich in CD56bright CD16⁻ NK cells. These cells contribute to tissue remodeling and immunological modulation by secreting substantial amounts of immunoregulatory cytokines as TNF-α and IFN-γ, but are less cytotoxic [54].
Early in pregnancy, NK cells also known as decidual NK (dNK) cells or uterine NK (uNK) cells are the main immune population in the uterus. They exhibit a CD56bright CD16⁻ phenotype, which is consistent with their important functions in tissue remodeling, immunological tolerance, and angiogenesis as compared with cytotoxicity. NK cells extracted from menstrual blood are occasionally referred to as eNK cells in the non- pregnant endometrium, whereas dNK cells develop from decidual tissue during pregnancy [40]. The developmental origin of uNK cells is still under investigation. Evidence suggests contributions from both circulating CD34+ hematopoietic progenitor cells (HPCs) and uterine- resident stem/progenitor cells. CD34+ precursors capable of differentiating into NK cells under IL-15 stimulation have been identified in bone marrow and uterine tissue. Furthermore, under the impact of cytokines like TGF-β, peripheral blood NK (pbNK) cells have demonstrated the capacity to develop decidual-like traits, underscoring the adaptability of NK cells differentiation in the uterine microenvironment. The function of circulating progenitors in repopulating the uNK compartment is further supported by data from HLA-mismatched uterine transplants [26].
5.2 Impact of the Tumor Microenvironment on NK Cells in Endometrial Cancer
Natural killer (NK) cells are essential parts of the innate immune system because they can identify and destroy cancerous or aberrant cells without being sensitized beforehand. By engaging inhibitory receptors such as killer-cell immunoglobulin-like receptors (KIRs) and NKG2A with self-MHC class I molecules, NK cells become functionally competent. This process is known as “education” or “licensing,” and it shapes their functioning. Without these signals, unlicensed NK cells are hyporesponsive in steady-state situations but can be triggered by cytokines such as IL-2, IL-15, and IL-18 in inflammatory settings, which lowers their activation threshold and increases cytotoxicity [18, 19]. NK cells kill target cells primarily via two mechanisms (Figure 9): granule-mediated exocytosis, releasing perforin, granzymes, Fas ligand (FasL), and TRAIL, which induce apoptosis; and death receptor-mediated killing through FasL and TRAIL interactions that activate caspase pathways [53, 54, 56].
Figure 9. Mechanisms by which NK Cells Identify and Eliminate Tumor Cells. Three different mechanisms are involved in the recognition of tumor cells: (1) the summation of signals from activating and inhibitory receptors; (2) the interaction of CD16 on the NK cell with immunoglobulin on the tumor cell surface; and (3) the interaction of the ligand (FasL) on the NK cell with the tumor cell's death receptor. Two mechanisms lead to the death of tumor cells: (1) the establishment of an immunological synapse, and (2) the start of death receptor-mediated apoptosis [54].
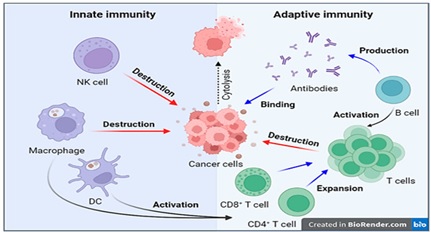
Additionally, NK cells bind to antibody-coated tumor cells via their CD16 receptor and induce antibody-dependent cellular cytotoxicity (ADCC). Despite being clinically utilized in a number of malignancies, ADCC’s function in EC is still poorly understood [53].
However, the tumor microenvironment (TME) in EC involves a variety of immune evasion techniques that significantly reduce the activity of NK cells. These include immunosuppressive enzymes, including indoleamine 2,3-dioxygenase (IDO) and B7-H4, downregulation of MHC class I molecules, and overexpression of immunological checkpoints like PD-L1. NK cytotoxicity is further hampered by metabolic problems in the TME, including hypoxia, nutritional depletion, and acidic pH [11, 53]. The functionally exhausted phenotype of tumor-infiltrating NK cells in EC is typified by decreased production of important cytotoxic mediators like IFN-γ, TNF-α, granzyme B, and CD107a, as well as increased expression of exhaustion markers (such as TIM-3). By interfering with IL-2 signaling, elevated levels of transforming growth factor-beta (TGF-β) in the TME reduce the cytotoxic capability of NK cells and decrease their mitochondrial metabolism. NK cells metabolic pathways, essential to their effector actions, are further inhibited by hypoxia and lactate buildup [27]. M2- polarized macrophages and myeloid-derived suppressor cells (MDSCs), which generate arginase-1 and nitric oxide via inducible nitric oxide synthase (iNOS), are among the immunosuppressive cells that are abundant in the EC microenvironment. This suppresses NK cells antibody- dependent cytotoxicity and general immune surveillance [27]. Additionally, altered cytokine and chemokine profiles such as decreased levels of chemoattractants CCL21 and CCL27 and increased immunosuppressive IL-6 impair NK cells recruitment and trafficking. Overexpression of CXCR4 by tumor cells and excessive consumption of its ligand CXCL12 reduce NK chemotaxis, sustaining the immunosuppressive milieu. Reduced CCL27 levels also contribute to impaired NK infiltration [10, 27].
In a mouse xenograft model, Yoshida et al., showed that tumors overexpressing IDO grew faster and were associated with lower splenic NK cells counts and cytotoxicity [57]. Treatment combining an IDO inhibitor with paclitaxel improved survival and tumor control. Additionally, progesterone-induced blocking factor (PIBF) may generate immunomodulatory proteins that suppress NK cytotoxicity, though its specific role in EC warrants further study [11]. Clinical studies have demonstrated significantly reduced NK cytotoxic activity in early-stage EC patients compared to healthy controls. This reduction correlates with adverse histopathological features, including higher tumor grade and deeper myometrial invasion. Garzetti et al., also found a negative correlation between NK function and nuclear grading, as well as PCNA proliferation marker expression [58]. Interestingly, the prognostic significance of NKp46+ NK cells infiltration appears dependent on HLA-E expression, where upregulation correlates with better outcomes, and normal expression predicts a poorer prognosis [11]. An unusual combination of uterine carcinosarcoma and metastatic invasive lobular carcinoma (ILC) of the breast was reported in a recent case report by Riedinger et al. [59]. Immunophenotyping showed that granzyme B+ and perforin+ cytotoxic NK cells, as opposed to the tissue-resident NK cells characteristic of type II EC, dominated the tumor immune microenvironment. In aggressive gynecologic malignancies, this implies that checkpoint inhibitor therapy may effectively rewire the NK cells landscape, restoring potent antitumor cytotoxicity [59]. These findings highlight the NK cells immunotherapeutic potential and functional complexity in EC. Novel immunotherapies that target the endometrial tumor microenvironment may be developed by utilizing their cytotoxic mechanisms and altering their receptor signaling pathways.
6. NK Cell Receptors and Their Functions in Endometrial Cancer
6.1 Diversity of NK Cells Receptors and Subsets
A wide range of receptors on natural killer (NK) cells enables them to identify and destroy aberrant or cancerous cells, including those found in EC (Table 3).
| Activating | Inhibitory |
| NKG2D ↓ | KIR2DL 1,2,3 |
| NKG2C | KIR2DL5 |
| FcyRIII | KIR3DL1 |
| NKp30 ↓ | LILRB1 |
| NKp44 | NKG2A ↑ |
| NKp46 ↓ | LAG-3 |
| NKp80 | KLRG1 |
| DNAM-1 ↑ | PD-1 ↑ |
| 2B4 (both of these are activating or inhibitory, depending on the stage.) | TIM-3 |
| CRACC (CD2-like receptor-activating cytotoxic cell) | Siglec-3 |
| CRTAM (Class I- restricted T cell–associated molecule) | Siglec - 7 |
| KIR2DS4 | Siglec -9 |
| KIR2DL4 | TIGIT ↑ |
| KIR3DS1 | IAP |
| NKRP1A | |
| LAIR-1 | |
| IRp60 | |
| Tactile | |
| IL1R8 |
Numerous NK cells receptors have been discovered, and the list is still growing (Figure 10) [49].
Figure 10. NK Cells and CD8 T Cells have Specific Activating and Inhibitory Receptors, Along with the Ligands that go with Them. NK cells activity is controlled by the balance of signals from cell surface activating and inhibitory receptors. On the other hand, T cell signaling is mostly mediated by the TCR, a master activating receptor, along with a number of co-stimulatory and co-inhibitory receptors. Select inhibitory receptors (-) are shown as a red negative signal, whereas select activating receptors (+) are shown as a green positive signal [60].
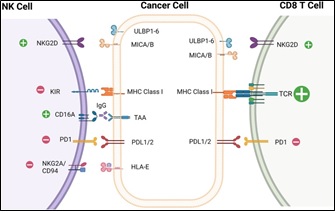
These receptors’ stochastic expression results in diverse NK cells subsets with unique functional abilities. The balance between activating and inhibitory receptors determines NK cells activity [54]. Activating receptors include the FcγRIIIA receptor CD16, which mediates antibody-dependent cellular cytotoxicity (ADCC); killer immunoglobulin-like receptors (KIRs), such as KIR- 2DS and KIR-3DS; C-type lectin-like receptors, such as NKG2D and NKG2C/CD94; and co-receptors, such as DNAM-1, 2B4, and NKp80, that amplify activation signals [27]. In contrast, immune evasion is facilitated by the upregulation of inhibitory receptors, including KIR-2DL, KIR-3DL, and CD94/NKG2A, as well as immune checkpoint molecules like PD-1, TIM-3, TIGIT, CD96, LAIR-1, and Siglec-7 inside the EC tumor microenvironment (Figure 11) [18, 27].
Figure 11. Human Natural Killer Cell Receptors of Interest. Yellow-highlighted activating and blue-highlighted inhibitory receptors are found on the surfaces of (A) pNK and (B) uNK, as well as the ligands that attach to them [49].
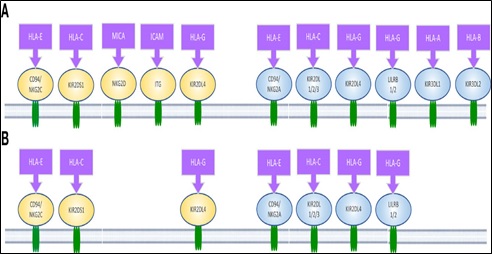
Immunoreceptor tyrosine-based activation motif (ITAM)-containing adaptor proteins mediate activating receptor signaling, which results in granule exocytosis and calcium mobilization. Conversely, inhibitory receptors communicate via immunoreceptor tyrosine-based inhibitory motifs (ITIMs), which block the activation of NK cells by attracting phosphatases that target important signaling molecules like Vav1, PLCγ, and LAT [18]. In order to initiate effector activities, NK cells need a net positive signal from these receptor connections. Both classical and non-classical MHC class I molecules, often known as human leukocyte antigens (HLAs), are NK receptor ligands [53].
The “missing-self” detection mechanism is mediated by inhibitory NK receptors, which enable NK cells to identify and destroy cells that exhibit decreased MHC-I expression, a characteristic frequently found in virus- infected or altered cells [53].
6.2 Targeting KIR and NKG2A to Restore NK Cells Function in Endometrial Cancer
Given the centrality of NK cells dysfunction in EC, particular attention has focused on the inhibitory receptors KIR and NKG2A, which represent the dominant checkpoints curbing NK cytotoxicity. These molecules not only mediate tolerance to self but are co-opted by tumor cells to evade immune elimination, underscoring their importance as therapeutic targets in EC.
The type I transmembrane glycoprotein family known as killer immunoglobulin-like receptors (KIRs) is mostly expressed on NK cells and a subset of T cells. Depending on their cytoplasmic patterns, these receptors can either provide activating or inhibitory signals in response to certain motifs on classical MHC class I molecules (HLA-A, -B, and -C). Immunoreceptor tyrosine-based inhibitory motifs (ITIMs) in the long cytoplasmic tails of inhibitory KIRs, including KIR2DL1-3, KIR2DL5, and KIR3DL1-3, reduce NK cells cytotoxicity when they interact with self-HLA ligands (Figure 12) [27].
Figure 12. Inhibitory Interactions of NK Cells with EC Cells Through KIR/HLA-C and NKG2A/HLA-E Axes. Created with BioRender.com (accessed on 4 November 2025).
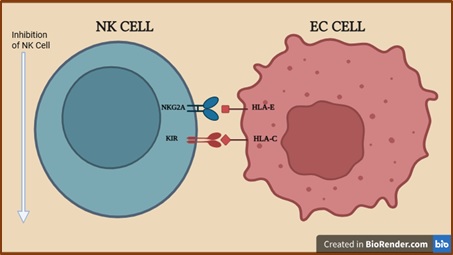
Individual differences in KIR gene expression lead to a variety of receptor-ligand interactions that affect the functional competence and education of NK cells. NK cells’ licensing status is determined by the interplay between the KIR and HLA genes, even though they are inherited separately; those that express inhibitory receptors that recognize self-HLA are regarded as functionally licensed [27]. Immune surveillance may be impaired in the case of EC due to changes in KIR expression or HLA ligand availability. For example, endometriosis, a disease epidemiologically connected to an elevated risk of EC, has been linked to decreased expression of KIR2DS2, an activating receptor that detects HLA-C1 and HLA-A [27]. Within the KIR family, KIR3DL3 is a unique inhibitory receptor that is incapable of binding traditional MHC class I molecules. Rather, it engages with HHLA2, a member of the B7 family that is overexpressed in a number of tumors (sometimes referred to as B7-H7). KIR3DL3 is abundant on the cytotoxic CD56^dim CD16+ subgroup, whereas TMIGD2 (CD28H), a co-stimulatory receptor, is mostly expressed on CD56^bright CD16⁻ NK cells. By interacting with KIR3DL3, tumor production of HHLA2 may prevent NK cells cytotoxicity; however, blocking this connection improves NK-mediated tumor clearance in vivo [55].
While direct evidence of HHLA2 expression in endometrial cancer (EC) is currently lacking, genomic and immunologic profiling by Levine et al., revealed that p53-abnormal and copy-number-high EC subtypes exhibit a profoundly immunosuppressive tumor microenvironment [12]. HHLA2, identified by Zhao et al., as a novel B7 family immune checkpoint, inhibits activation of both CD4⁺ and CD8⁺ T cells [62], suggesting that HHLA2-mediated inhibitory signaling well documented in other malignancies could plausibly contribute to immune evasion in aggressive EC subtypes with high genomic instability. In other tumors, including breast, lung, and osteosarcoma, high HHLA2 expression is associated with increased tumor aggressiveness, immune evasion, and poorer prognosis [63], supporting the idea that HHLA2 may similarly function as an immune evasion mechanism in EC. Thus, the KIR3DL3-HHLA2 axis represents a novel immunological checkpoint and potential therapeutic target; however, its precise role in EC remains to be elucidated, and further research is warranted to clarify how KIR3DL3 and TMIGD2 interactions influence NK cell activity [55].
NKG2A is a C-type lectin-like inhibitory receptor that binds to the non-classical MHC class I protein HLA-E and forms a heterodimer with. The receptor is found on mature fractions of CD56-bright NK cells and is highly expressed on immature cells, particularly in tumor-associated or chronic inflammatory situations. NKG2A inhibits NK cells cytotoxicity and cytokine production by transmitting inhibitory signals through ITIMs upon interaction with HLA-E, which is frequently increased on tumor cells [18]. By identifying HLA-E molecules presenting leader sequence-derived peptides from classical MHC class I molecules like HLA-G, the CD94/NKG2A heterodimer acts as a dominant inhibitory receptor (Figure 12). With a sixfold greater affinity than the activating CD94/NKG2C receptor, this connection allows NK cells to distinguish between activating and inhibitory inputs accurately [27].
In EC, aberrant HLA-E expression contributes to impaired NK cells-mediated immune responses by disrupting the “missing-self” recognition mechanism. Studies have demonstrated that upregulation of CD94/ NKG2A on NK cells correlates with diminished cytotoxicity in EC and related conditions like endometriosis. For example, Galandrini et al., reported that peritoneal NK cells from endometriosis patients expressed high levels of CD94/NKG2A and were unable to lyse HLA-E–expressing target cells unless this interaction was blocked by anti-NKG2A antibodies, thereby restoring cytotoxic activity [64]. NKG2A is expressed early during NK cells development and remains predominant on CD56^bright NK cells, which are cytokine-producing and less cytotoxic. In contrast, CD56^dim NK cells exhibit stronger lytic function and co-express NKG2A along with KIRs, allowing precise tuning of effector responses. Tumor cells frequently exploit this inhibitory axis by overexpressing HLA-E or inducing immunosuppressive cytokines like TGF-β, both of which dampen NK cells activity and promote functional exhaustion [18].
Within the tumor microenvironment, this inhibitory pathway serves as a potent mechanism of immune evasion. Although HLA-E is constitutively transcribed in many tissues, its surface expression on tumor cells is often variable and inducible by immune cytokines such as IFN-γ. This cytokine-driven upregulation can enhance NKG2A engagement and suppress NK cells responses [18]. Interestingly, some studies suggest a paradox where elevated HLA-E expression is associated with improved prognosis in certain cancers. This may reflect a context-dependent interplay between inhibitory NKG2A and activating NKG2C receptors, influenced by the distribution and activation status of NK subsets and the tumor cytokine milieu [26]. Functionally, KIRs and NKG2A function as important immunological checkpoints for NK cells, much like PD-1 and CTLA-4 do for T cells. However, KIRs and NKG2A are constitutively produced at steady state to preserve NK cells tolerance to self under physiological settings, in contrast to T cell checkpoints, which are increased post-activation and linked to exhaustion [55].
In conclusion, Natural killer (NK) cells play a pivotal role in the immunological surveillance of endometrial cancer (EC) through their ability to recognize and destroy malignant cells independently of classical MHC restriction. Compelling evidence discussed in this review indicates that the inhibitory receptors KIR and NKG2A, together with their ligands HLA-C and HLA-E, act as dominant innate immune checkpoints that impair NK-cell cytotoxicity within the immunosuppressive tumor microenvironment of EC. Dysregulation of these pathways undermines NK- cell licensing, cytokine production, and effector function, thereby facilitating immune escape and tumor progression. This review highlights a novel and complementary immunotherapeutic perspective: that targeting innate immune checkpoints such as KIR and NKG2A may enhance the efficacy of existing T-cell-directed checkpoint inhibitors, particularly in specific molecular EC subtypes (e.g., MSI-H and POLE-mutated tumors) characterized by heightened immunogenicity. Future research should focus on delineating the heterogeneity of KIR haplotypes and HLA-E expression across EC molecular subtypes, investigating combinatorial blockade strategies involving anti-NKG2A and PD-1/PD-L1 inhibitors, and exploring predictive biomarkers to stratify patients most likely to benefit from NK-cell–based immunotherapy in EC.
Acknowledgments
Statement of Transparency and Principles
• The authors declare no conflict of interest.
• The study was approved by the Research Ethics Committee of the authors’ affiliated institution.
• The study data are available upon reasonable request.
• All authors contributed to the implementation of this research.
References
- Intraoperative Flow Cytometry for the Characterization of Gynecological Malignancies Anastasiadi Z, Mantziou S, Akrivis C, Paschopoulos M, Balasi E, Lianos GD , Alexiou GA , et al . Biology.2022;11(9). CrossRef
- Molecular mechanism of tumor-infiltrating immune cells regulating endometrial carcinoma Ding S, Hao Y, Qi Y, Wei H, Zhang J, Li H. Genes & Diseases.2025;12(3). CrossRef
- Recent Advances in Endometrial Cancer Prevention, Early Diagnosis and Treatment Baker-Rand H, Kitson SJ . Cancers.2024;16(5). CrossRef
- Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries Sung H, Ferlay J, Siegel RL , Laversanne M, Soerjomataram I, Jemal A, Bray F. CA: A Cancer Journal for Clinicians.2021;71(3). CrossRef
- Immune Environment and Immunotherapy in Endometrial Carcinoma and Cervical Tumors Lainé A, Gonzalez-Lopez AM , Hasan U, Ohkuma R, Ray-Coquard I. Cancers.2023;15(7). CrossRef
- Variations in incidence and mortality rates of endometrial cancer at the global, regional, and national levels, 1990–2019 Gu B, Shang X, Yan M, Li X, Wang W, Wang Q, Zhang C. Gynecologic Oncology.2021;161(2). CrossRef
- Cancer Research UK. Uterine Cancer Statistics. Available at: https://www.cancerresearchuk.org/health-professional/cancerstatistics/statistics-by-cancer-type/uterine-cancer#heading-Six .
- SEER. Cancer of the Endometrium - Cancer Stat Facts.. Available at: https://seer.cancer.gov/statfacts/html/corp.html .
- Two pathogenetic types of endometrial carcinoma Bokhman JV . Gynecologic Oncology.1983;15(1). CrossRef
- Tissue Infiltrating Immune Cells and Endometrial Cancer Prognosis Drakes M, Mehrotra SK , Potkul RM , Czerlanis CJ , Stiff P. Clinical Oncology and Research.2021. CrossRef
- Immune disorder in endometrial cancer: Immunosuppressive microenvironment, mechanisms of immune evasion and immunotherapy (Review) Zhan L, Liu X, Zhang J, Cao Y, Wei B. Oncology Letters.2020;20(3). CrossRef
- Integrated genomic characterization of endometrial carcinoma Levine DA . Nature.2013;497(7447). CrossRef
- ESGO/ESTRO/ESP guidelines for the management of patients with endometrial carcinoma Concin N, Matias-Guiu X, Vergote I, Cibula D, Mirza MR , Marnitz S, et al . International Journal of Gynecological Cancer.2021;31(1). CrossRef
- Tumor immune microenvironment in endometrial cancer of different molecular subtypes: evidence from a retrospective observational study Dai Y, Zhao L, Hua D, Cui L, Zhang X, Kang N, et al . Frontiers in Immunology.2022;13. CrossRef
- Importance of the endometrial immune environment in endometrial cancer and associated therapies Van Der Woude H, Hally KE , Currie MJ , Gasser O, Henry CE . Frontiers in Oncology.2022;12. CrossRef
- Features of the immunosuppressive tumor microenvironment in endometrial cancer based on molecular subtype Zhang C, Wang M, Wu Y. Frontiers in Oncology.2023;13. CrossRef
- CD8 T Cell Exhaustion During Chronic Viral Infection and Cancer McLane LM , Abdel-Hakeem MS , Wherry EJ . Annual Review of Immunology.2019;37(1). CrossRef
- Key Activating and Inhibitory Ligands Involved in the Mobilization of Natural Killer Cells for Cancer Immunotherapies Karmakar S, Pal P, Lal G. ImmunoTargets and Therapy.2021;Volume 10. CrossRef
- Endometrial Tumor Microenvironment Alters Human NK Cell Recruitment, and Resident NK Cell Phenotype and Function Degos C, Heinemann M, Barrou J, Boucherit N, Lambaudie E, Savina A, Gorvel L, Olive D. Frontiers in Immunology.2019;10. CrossRef
- A clinically applicable molecular-based classification for endometrial cancers Talhouk A, McConechy M K, Leung S, Li-Chang H H, Kwon J S, Melnyk N, et al . British Journal of Cancer.2015;113(2). CrossRef
- Improved Risk Assessment by Integrating Molecular and Clinicopathological Factors in Early-stage Endometrial Cancer—Combined Analysis of the PORTEC Cohorts Stelloo E, Nout RA , Osse EM , Jürgenliemk-Schulz IJ , Jobsen JJ , Lutgens LC , Van Der Steen-Banasik EM , et al . Clinical Cancer Research.2016;22(16). CrossRef
- Classification of high-grade endometrium carcinomas using molecular and immunohistochemical methods Bayramoglu D, Seçilmiş Kerimoğlu Ö, Bayramoğlu Z, Çintesun E, Şahin G, Karabağlı P, Çelik Ç. Ginekologia Polska.2023;94(1). CrossRef
- Response to lenvatinib and pembrolizumab combination therapy in pembrolizumab-pretreated relapsed endometrial cancer Mimura K, Shimomura A, Gota T, Ando K, Kawamura Y, Taniyama T, Oishi H, Shimizu C. Gynecologic Oncology Reports.2022;44. CrossRef
- TCGA Molecular Prognostic Groups of Endometrial Carcinoma: Current Knowledge and Future Perspectives Arciuolo D, Travaglino A, Raffone A, Raimondo D, Santoro A, Russo D, Varricchio S, et al . International Journal of Molecular Sciences.2022;23(19). CrossRef
- Molecular Classification of Endometrial Cancer and Its Impact on Therapy Selection Galant N, Krawczyk P, Monist M, Obara A, Gajek Ł, Grenda A, Nicoś M, Kalinka E, Milanowski J. International Journal of Molecular Sciences.2024;25(11). CrossRef
- Biology and pathology of the uterine microenvironment and its natural killer cells Wang F, Qualls AE , Marques-Fernandez L, Colucci F. Cellular & Molecular Immunology.2021;18(9). CrossRef
- Chasing Uterine Cancer with NK Cell-Based Immunotherapies Kumar V, Bauer C, Stewart JH . Future Pharmacology.2022;2(4). CrossRef
- C-Reactive Protein as a Prognostic Biomarker for Gynecologic Cancers: A Meta-Analysis Yang Y, Li X, Qian H, Di G, Zhou R, Dong Y, Chen W, Ren Q. Computational Intelligence and Neuroscience.2022;2022. CrossRef
- CTHRC1 promotes M2-like macrophage recruitment and myometrial invasion in endometrial carcinoma by integrin-Akt signaling pathway Li L, Yin K, Bai Y, Zhang Z, Di W, Zhang S. Clinical & Experimental Metastasis.2019;36(4). CrossRef
- Long non-coding RNA NIFK-AS1 inhibits M2 polarization of macrophages in endometrial cancer through targeting miR-146a Zhou Y, Zhao W, Mao L, Wang Y, Xia L, Cao M, Shen J, Chen J. The International Journal of Biochemistry & Cell Biology.2018;104. CrossRef
- CD8 down-regulation on cytotoxic T lymphocytes of patients with endometrioid endometrial carcinomas Pascual-García M, Bértolo C, Nieto JC , Serrat N, Espinosa Í, D'Angelo E, Muñoz R, et al . Human Pathology.2016;56. CrossRef
- Intratumoral CD8+ T Lymphocytes as a Prognostic Factor of Survival in Endometrial Carcinoma Kondratiev S, Sabo E, Yakirevich E, Lavie O, Resnick MB . Clinical Cancer Research.2004;10(13). CrossRef
- Intratumoral CD8+ Lymphocyte Infiltration as a Prognostic Factor and Its Relationship With Cyclooxygenase 2 Expression and Microsatellite Instability in Endometrial Cancer Suemori T, Susumu N, Iwata T, Banno K, Yamagami W, Hirasawa A, Sugano K, Matsumoto E, Aoki D. International Journal of Gynecological Cancer.2015;25(7). CrossRef
- Prognostic implication of programmed cell death 1 protein and its ligand expressions in endometrial cancer Kim J, Kim S, Lee HS , Yang W, Cho H, Chay DB , Cho SJ , Hong S, Kim J. Gynecologic Oncology.2018;149(2). CrossRef
- The immunologic tumor microenvironment in endometrioid endometrial cancer in the morphomolecular context: mutual correlations and prognostic impact depending on molecular alterations Willvonseder B, Stögbauer F, Steiger K, Jesinghaus M, Kuhn P, Brambs C, Engel J, et al . Cancer Immunology, Immunotherapy.2021;70(6). CrossRef
- Clinical significance of regulatory T cells and CD8+ effector populations in patients with human endometrial carcinoma Chang W, Li C, Huang S, Chang D, Chou L, Sheu B. Cancer.2010;116(24). CrossRef
- Immunofluorescence-Detected Infiltration of CD4+FOXP3+ Regulatory T Cells is Relevant to the Prognosis of Patients with Endometrial Cancer Yamagami W, Susumu N, Tanaka H, Hirasawa A, Banno K, Suzuki N, Tsuda H, Tsukazaki K, Aoki D. International Journal of Gynecological Cancer.2011;21(9). CrossRef
- Immunosuppressive Tumor Microenvironment Status and Histological Grading of Endometrial Carcinoma Antomarchi J, Ambrosetti D, Cohen C, Delotte J, Chevallier A, Karimdjee-Soilihi B, Ngo-Mai M, Schmid-Alliana A, Schmid-Antomarchi H. Cancer Microenvironment.2019;12(2-3). CrossRef
- Tumor-Infiltrated Lymphocytes, Macrophages, and Dendritic Cells in Endometrioid Adenocarcinoma of Corpus Uteri as Potential Prognostic Factors Zinovkin D, Pranjol MZI . International Journal of Gynecological Cancer.2016;26(7). CrossRef
- Endometrial Cancer Immune Escape Mechanisms: Let Us Learn From the Fetal–Maternal Interface Bruno V, Corrado G, Baci D, Chiofalo B, Carosi MA , Ronchetti L, Piccione E, et al . Frontiers in Oncology.2020;10. CrossRef
- Prognostic significance of tumor-associated macrophages in endometrial adenocarcinoma Kübler K, Ayub TH , Weber SK , Zivanovic O, Abramian A, Keyver-Paik M, Mallmann MR , et al . Gynecologic Oncology.2014;135(2). CrossRef
- Tumor-associated macrophages correlate with vascular space invasion and myometrial invasion in endometrial carcinoma Soeda S, Nakamura N, Ozeki T, Nishiyama H, Hojo H, Yamada H, Abe M, Sato A. Gynecologic Oncology.2008;109(1). CrossRef
- Infiltration of Tumor-associated Macrophages is Increased in the Epithelial and Stromal Compartments of Endometrial Carcinomas: Dun EC , Hanley K, Wieser F, Bohman S, Yu J, Taylor RN . International Journal of Gynecological Pathology.2013;32(6). CrossRef
- Detection of Lymphovascular Invasion by D2-40 (Podoplanin) Immunoexpression in Endometrial Cancer Weber SK , Sauerwald A, Pölcher M, Braun M, Debald M, Serce NB , Kuhn W, Brunagel-Walgenbach G, Rudlowski C. International Journal of Gynecological Cancer.2012;22(8). CrossRef
- Exploring the Immunological Mechanisms Underlying the Anti-vascular Endothelial Growth Factor Activity in Tumors Aguiar RBD , Moraes JZD . Frontiers in Immunology.2019;10. CrossRef
- Intratumoral neutrophils and plasmacytoid dendritic cells indicate poor prognosis and are associated with pSTAT3 expression in AJCC stage I/II melanoma Jensen TO , Schmidt H, Møller HJ , Donskov F, Høyer M, Sjoegren P, Christensen IJ , Steiniche T. Cancer.2012;118(9). CrossRef
- Tumor-Infiltrating Dendritic Cells May Be Used as Clinicopathologic Prognostic Factors in Endometrial Carcinoma Lijun Z, Xin Z, Danhua S, Xiaoping L, Jianliu W, Huilan W, Lihui W. International Journal of Gynecological Cancer.2012;22(5). CrossRef
- Endometrial Carcinoma: Immune Microenvironment and Emerging Treatments in Immuno-Oncology Rousset-Rouviere S, Rochigneux P, Chrétien A, Fattori S, Gorvel L, Provansal M, Lambaudie E, Olive D, Sabatier R. Biomedicines.2021;9(6). CrossRef
- Relative expression of receptors in uterine natural killer cells compared to peripheral blood natural killer cells Ismail NI . Frontiers in Immunology.2023;14. CrossRef
- Loss of HLA class I and mismatch repair protein expression in sporadic endometrioid endometrial carcinomas De Jong RA , Boerma A, Boezen HM , Mourits MJE , Hollema H, Nijman HW . International Journal of Cancer.2012;131(8). CrossRef
- Characterization of human leukocyte antigen-G (HLA-G) expression in endometrial adenocarcinoma Barrier BF , Kendall BS , Sharpe-Timms KL , Kost ER . Gynecologic Oncology.2006;103(1). CrossRef
- Immunoexpression of B7-H3 in endometrial cancer: Relation to tumor T-cell infiltration and prognosis Brunner A, Hinterholzer S, Riss P, Heinze G, Brustmann H. Gynecologic Oncology.2012;124(1). CrossRef
- Natural Killer Cell-Based Immunotherapy in Gynecologic Malignancy: A Review Uppendahl LD , Dahl CM , Miller JS , Felices M, Geller MA . Frontiers in Immunology.2018;8. CrossRef
- Antitumor Immunity: Role of NK Cells and Extracellular Vesicles in Cancer Immunotherapy Prokopeva AE , Emene CC , Gomzikova MO . Current Issues in Molecular Biology.2023;46(1). CrossRef
- Inhibitory Receptors and Immune Checkpoints Regulating Natural Killer Cell Responses to Cancer Buckle I, Guillerey C. Cancers.2021;13(17). CrossRef
- Single-cell transcriptomics reveals comprehensive microenvironment and highlights the dysfuntional state of NK cells in endometrioid carcinoma Shi W, Wu W, Wang J, Meng X. Medicine.2024;103(13). CrossRef
- Overexpression of Indoleamine 2,3-Dioxygenase in Human Endometrial Carcinoma Cells Induces Rapid Tumor Growth in a Mouse Xenograft Model Yoshida N, Ino K, Ishida Y, Kajiyama H, Yamamoto E, Shibata K, Terauchi M, et al . Clinical Cancer Research.2008;14(22). CrossRef
- Natural Killer Cell Activity in Stage I Endometrial Carcinoma: Correlation with Nuclear Grading, Myometrial Invasion, and Immunoreactivity of Proliferating Cell Nuclear Antigen Garzetti GG , Ciavattini A, Goteri G, Tranquilli AL , Muzzioli M, Fabris N, De Nictolis M, Romanini C. Gynecologic Oncology.1994;55(1). CrossRef
- Case report: Characterization of the immunologic and molecular landscape in a unique presentation of invasive lobular carcinoma with concurrent uterine carcinosarcoma treated with immunotherapy Riedinger CJ , Eisele CD , Esnakula A, Stover DG , Freud AG , Cosgrove CM . Frontiers in Immunology.2024;15. CrossRef
- Engaging natural killer cells for cancer therapy via NKG2D, CD16A and other receptors Whalen KA , Rakhra K, Mehta NK , Steinle A, Michaelson JS , Baeuerle PA . mAbs.2023;15(1). CrossRef
- NK cells in uterine cancers. LSU Health Digital Scholar Chang S, Kumar V, Bauer C, Stewart H. 2022. https:// digitalscholar.lsuhsc.edu/sommrd/2022MRD/Posters/14/.
- HHLA2 is a member of the B7 family and inhibits human CD4 and CD8 T-cell function Zhao R, Chinai JM , Buhl S, Scandiuzzi L, Ray A, Jeon H, Ohaegbulam KC , et al . Proceedings of the National Academy of Sciences.2013;110(24). CrossRef
- The third group of the B7-CD28 immune checkpoint family: HHLA2, TMIGD2, B7x, and B7-H3 Janakiram M, Shah UA , Liu W, Zhao A, Schoenberg MP , Zang X. Immunological Reviews.2017;276(1). CrossRef
- Increased frequency of human leukocyte antigen–E inhibitory receptor CD94/NKG2A–expressing peritoneal natural killer cells in patients with endometriosis Galandrini R, Porpora MG , Stoppacciaro A, Micucci F, Capuano C, Tassi I, Di Felice A, Benedetti-Panici P, Santoni A. Fertility and Sterility.2008;89(5). CrossRef
Content
Abstract 1. Introduction 2. Molecular Classification of Endometrial Cancer 3. Immune Microenvironment of the Normal Endometrium: Foundation for Understanding EC Immunity 4. Endometrial Cancer’s Immune Microenvironment and Immune Escape Mechanisms 5. Uterine Natural Killer (uNK) Cells 6. NK Cell Receptors and Their Functions in Endometrial Cancer Acknowledgments ReferencesLicense

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
Copyright
© Asian Pacific Journal of Cancer Biology , 2026
Author Details