Synergistic Cytotoxicity of Newcastle Disease Virus and High-Dose Dimethyl Fumarate in Breast Cancer Cells Is Accompanied by reduction of Selectivity in Normal Cells
Download
Abstract
Background: Newcastle disease virus (NDV) is a promising oncolytic virus with selective cytotoxicity against cancer cells, while dimethyl fumarate (DMF) is an FDA-approved NF-κB inhibitor showing interesting anticancer activity. The current work assessed the combined cytotoxic effect of NDV and DMF on breast cancer cells and on normal rat embryo fibroblast (REF) cells.
Methods: Killing ability was evaluated using crystal violet cytotoxicity assay following treatment with NDV (MOI 1–5), DMF (15.6–500 µg/mL), or their combinations. Drug interaction was measured using the Chou–Talalay combination index.
Results: NDV alone showed higher cytotoxicity toward breast cancer cells than toward normal REF cells, confirming its known selectivity. DMF demonstrated dose-dependent cytotoxicity in all tested cell lines. Interestingly, combining NDV with medium–high concentrations of DMF produced strong synergistic effects not only in breast cancer cells but also in normal REF cells. This indicates that DMF eliminates the essential cancer-selectivity of NDV and increases normal-cell susceptibility to the combination treatment.
Conclusion: While NDV and DMF display strong synergistic cytotoxicity in vitro, this synergy also induced in normal cells, resulting in a loss of the selective oncolytic nature of NDV. These findings emphasize a major limitation of the combination strategy and highlight the need for dose-optimization studies with lower DMF concentrations or alternative NF-κB inhibitors to preserve selectivity while maintaining anticancer activity.
Introduction
Since the discovery of the oncolytic properties of NDV in the 1950s, it has been extensively utilized in preclinical research as an innovative anticancer agent for various solid and resistant tumors, including breast, gastric, liver, lung, cervical, prostate, colorectal cancers, and glioblastoma [1]. Out of 53 viruses evaluated as anticancer treatments, 38 had antineoplastic effects in both animal and human subjects [2]. To be considered optimal, a virus must be oncolytic but nonpathogenic to humans. The list of such viruses is short and includes Newcastle disease virus (NDV) [3]. The Newcastle disease virus (NDV) is an avian paramyxovirus that can induce significant respiratory, gastrointestinal, and neurological diseases in chicken, resulting in economic detriment to the global poultry industry [4]. Notably, NDV has a strong affinity for cancerous cells, causing them to lyse while preserving undamaged healthy cells [5]. NDV’s tumor-specific replication makes it a desirable choice for oncolytic virotherapy. In addition to lysing tumor cells directly, NDV can cause necrosis, autophagy, inflammation, apoptosis, and immunogenic cell death by activating a number of signaling pathways [6]. Emerging research suggests that NDV has the ability to impair cancer cell metabolism. NDV, a promising treatment for breast cancer, can be applied to preclinical viral oncolytic medicines [7]. Breast cancer is a difficult-to-treat type of cancer because of its high recurrence rate, risk of metastasis, and chemoresistance. Therefore, there is a need for effective treatments that can overcome resistance to treatment and attack cancer cells via multiple therapeutic agents and utilize different mechanisms of cancer cell death [8]. NF-κB, a transcription factor that governs cell division, proliferation, and survival, is critical for cancer formation. Targeting NF-κB can improve the susceptibility of tumor cells to chemotherapy. The FDA-approved NF-κB inhibitor dimethyl fumarate (DMF) additionally displays promise in preventing the development of breast cancer cells in vitro [9]. Many reports have highlighted the activation of NF-κB and suggested that NF-κB plays a crucial role in increasing the proliferation of cells via the transcription of antiapoptotic proteins [10, 11]. DMF is a drug that is used therapeutically to treat moderate-to-severe psoriasis or relapsing-remitting multiple sclerosis [12]. DMF’s mode of action involves two processes that limit the activity of NF-κB, a transcription factor that regulates gene expression in inflammation and can stimulate Nrf2-dependent pathways. Nrf2 activation can reduce inflammatory responses by reducing the production of pro-inflammatory cytokines [13, 14]. The goal of the current work is to investigate the combined lethal impact of NDV oncolytic virotherapy and dimethyl fumarate against breast cancer, exploring the possible synergy in the elimination of cancer cells in vitro.
Materials and Methods
NDV propagation
The Experimental Therapy Department/Iraqi Center of Cancer and Medical Genetics Research (ICCMGR), Mustansiriyah University, Baghdad, Iraq, handed the Newcastle disease virus (Iraqi AMHA1 strain). A After puncturing the 10-day embryonated chicken eggs, 0.1 ml of attenuated NDV was propagated in them, and the eggs were covered with paraffin. Egg survival was checked daily in a 37°C humidified incubator. Following death, the embryo was promptly placed in a refrigerator. Within half an hour, The allantoic fluid was collected using a sterile syringe and then cleansed from debris by centrifugation (3000 rpm, 30 minutes at 4 °C). Following collection, a 0.45 mm filter was used to sterile the supernatant. The virus was transferred into small tubes and stored at-86°C.
Titration of NDV
For virus titration, we used a hemagglutination assay to determine the number of viruses. A healthy chicken’s brachial veins under its wing were pierced using a sterile needle. After that, the blood was transferred to an ethylenediaminetetraacetic acid (EDTA) tube and centrifuged three times at 4°C at 1000 RPM for 10 minutes. After the supernatant was discarded, 0.1 ml of the cells were transferred to a sterile tube. Finally, 1% of the chicken red blood cells were suspended in phosphate-buffered saline. The final virus titer was evaluated using a standard method that incorporates a 50% tissue culture infective dose (TCID50), with Vero cells titrated according to an established protocol.
Cell lines and culture
The Cell Bank Unit, Experimental Therapy Department, ICCMGR, Baghdad, Iraq, supplied the estrogen and progesterone receptor-positive MCF-7 human breast cancer cell line, the estrogen and progesterone receptor-negative AMJ13 human breast cancer cell line, and the standard rat embryo fibroblast line (REF). The AMJ13 and REF cell lines were grown in RPMI-1640 media supplemented with sodium bicarbonate (2.0 g/L) and l-glut (0.3 g/L). MCF-7 cells, on the other hand, were maintained in MEM (US Biological, USA) enriched with ten percent (v/v) fetal bovine serum (FBS) (Capricorn-Scientific, Germany) and 1% (v/v) penicillin-streptomycin (Capricorn-Scientific, Germany), and underwent the incubation process in a humidified environment of 5% CO2 at 37 °C, and cells that developed were used for the experiments.
Measurement of Cytotoxicity
In a 96-well microplate, cells were grown at a density of 1 × 104 per well and permitted to develop overnight at 37°C until an inverted microscope revealed a 60-70% confluence monolayer. NDV concentrations at diluted multiplicities of infection (MOIs) of 1, 3, and 5 were tested. After 2 hours, remove the media and add 100 µl of serum-free medium to each well. The mixture was then cultured for another 72 hours. DMF Pharmaceuticals (Almirall, S.A.) was purchased from the pharmacy. This agent was dissolved in RPMI-1640-free serum to obtain a stock solution before use for in vitro studies. The concentrations of diluted DMF were 500 µg/ml, 250 µg/ ml, 125 µg/ml, 62.5 µg/ml, 31.25 µg/ml and 15.6 µg/ml. For combinations of NDV with DMF, the MOI of NDV was 1, 3, and 5 while the concentrations of diluted DMF were 125 µg/ml, 62.5 µg/ml and 31.25 µg/ml, respectively. After two hours, the NDVs were discarded, and each well received 100 µl from different dilutions of DMF. After 72 hours, the medium was removed and 50 µl of a 1 mg/ml crystal violet dye solution (Bio-World, USA) was added to each well. After 20 minutes of incubation, the plates were gently washed with tap water to remove the stains. The cells have been photographed with a digital camera after being observed using an inverted microscope at 40× magnification. After adding 200 µl of methanol, the optical density of treated and control cells was measured using a 584 nm ELISA plate reader. The endpoint metric for each cell line was the inhibition rate of cell growth (cytotoxicity%), which was computed as follows:
Cytotoxicity% = OD Control − OD sample/OD Control × 100
where OD control is the average optical density of the untreated wells and OD sample is the average optical density of the treated wells [15].
Statistical analysis
The data were statistically analyzed using a one-way ANOVA approach in GraphPad Prism (GraphPad Software, California, USA). The standard deviation of the mean was significant at P = 0.05, whereas P < 0.05 was declared significant. Data in graphs are shown as means ±.S.D.
Combined cytotoxicity assays and Chou–Talalay analysis
The results of the cytotoxicity tests for IC50 determination were utilized to select the doses for this study. Concentrations of NDV and DMF were used for the AMJ13, MCF-7, and REF cell lines. Breast cancer cell lines and the REF normal cell line were transferred onto 96-well plates with a density of 1 × 10⁴ cells per well and kept in an incubator overnight. In the growth inhibition assay, NDV was introduced at MOIs of 1, 3, and 5, followed by serial dilutions of dimethyl fumarate at dosages of 500, 250, 125, 62.5, 31.25, and 15.6 μg/mL. After 72 hours of treatment, growth inhibition was assessed. To evaluate synergism, nonconstant ratio combinations of NDV and DMF were tested in triplicate. The Chou‒Talalay combination index (CI) was calculated via CompuSyn software (CompuSyn Inc., Paramus, NJ, USA) on the basis of mutually exclusive equations with unfixed ratios of NDV and DMF. The Chou-Talalay combination index (CI) was derived using CompuSyn software (CompuSyn Inc., Paramus, NJ, USA) using mutually exclusive equations with variable NDV and DMF ratios. We interpreted the CI values as follows: Additive effects range from 0.9 to 1.1, whereas synergistic effects are less than 0.9 and antagonistic effects exceed 1.1 [16].
Results
Cytotoxicity of DMF and NDV to breast cancer and normal cells
The effects of several doses of dimethyl fumarate (DMF) and multiplicities of infection (MOIs) of Newcastle disease virus (NDV) on several cell lines were evaluated by a cytotoxicity test. DMF cytotoxicity percentages for the AMJ-13 cell line (Figure 1), were 76.6% at 500 μg/ ml, 76.3% at 250 μg/ml, 75.6% at 125 μg/ml, 65.7% at 62.5 μg/ml, 63.8% at 31.25 μg/ml, and 58.3% at 15.6 μg/ml. With CT%, values of 45.7% for 5 MOI, 40% for 3 MOI, and 27% for 1 MOI, the cells were less sensitive to NDV virotherapy.
Figure 1. Cytotoxicity of DMF and NDV in AMJ-13 Cells. AMJ-13 breast cancer cells were treated with DMF (500, 250,125,62.5,31.25 and 15.6 μg/ml) or NDV at MOIs 1, 3, and 5 for 72 h. DMF produced dose-dependent cytotoxicity (IC50 = 22.45 µg/mL), whereas NDV showed moderate cytotoxicity with increasing MOI. Data represent mean ± SEM of three independent experiments. Statistical significance was determined using one-way ANOVA (*P < 0.05; **P < 0.01; ***P < 0.001). This figure establishes baseline monotherapy effects for subsequent combination analysis .
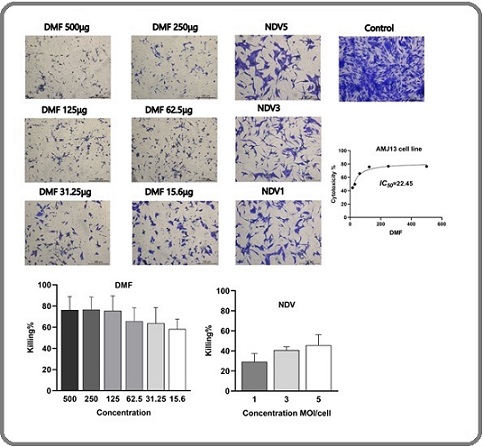
Calculated to be 22.45 μg/ml was the IC50 for the AMJ-13 cell line.
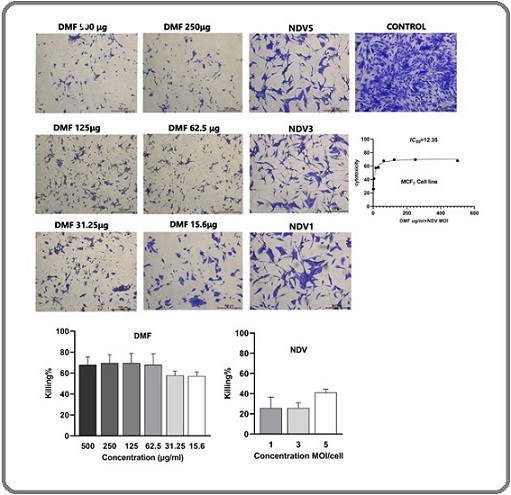
Another breast cancer cell line MCF-7 cell line (Figure 2), the cytotoxicity percentages of DMF were 68% at 500 μg/ml, 69% at 250 μg/ml, 69% at 125 μg/ml, 67% at 62.5 μg/ml, 58% at 31.25 μg/ml, and 57% at 15.6 μg/ml. In contrast, the CT% values for NDV virotherapy were lower: 41.2% at 5 MOI, 26% at 3 MOI, and 25% at 1 MOI. The IC50 was determined to be 12.35 μg/ml.
Figure 2. Cytotoxicity of DMF and NDV in MCF-7 Cells. MCF-7 cells were exposed to DMF (500, 250, 125, 62.5, 31.25 and 15.6 µg/mL) and NDV (MOI 1, 3, and 5) for 72 h. DMF showed strong cytotoxicity across all doses (IC50 = 12.35 µg/mL), while NDV induced lower, MOI-dependent cytotoxicity. Data represent mean ± SEM; *P < 0.05; **P < 0.01; ***P < 0.001. This figure displays that NDV monotherapy is less potent than DMF in this cell line.
As for the REF cell line normal rat embryo fibroblast cells (Figure 3), the cytotoxicity percentages of DMF were 70% at 500 μg/ml, 68% at 250 μg/ml, 65% at 125 μg/ml, 60% at 62.5 μg/ml, 58% at 31.25 μg/ml, and 29% at 15.6 μg/ml. Comparatively, the CT% values for NDV virotherapy were significantly lower, at 27% for 5 MOI, 23% for 3 MOI, and 19% for 1 MOI. The IC50 for the REF cell line was calculated as 18 μg/ml.
Figure 3. Cytotoxicity of DMF and NDV in Normal REF Cells. REF fibroblasts were treated with DMF (500, 250, 125, 62.5, 31.25 and 15.6 µg/mL) or NDV (MOI 1, 3, and 5). DMF displayed substantial cytotoxicity(IC50=18µg/mL),whileNDVconservedlower toxicity, confirming NDV’s natural selectivity toward cancer cells. Values represent mean ± SEM of triplicate experiments (*P < 0.05; **P < 0.01; ***P < 0.001).
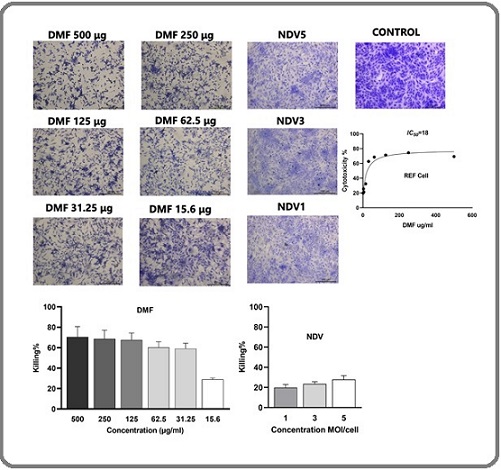
Throughout all three cell lines, NDV monotherapy demonstrated a graded cytotoxic effect, with stronger response in breast cancer cells than in REF cells, proving NDV’s known tumor selectivity. In contrast, high DMF doses produced similar cytotoxicity in both cancer and normal cells, indicating a narrower therapeutic window. This contrast between NDV and DMF monotherapies establishes the baseline needed to interpret the combination results.
The combined killing effect of DMF and NDV to breast cancer and normal cells
When NDV was combined with DMF, a clear boost of cytotoxicity was observed in all three cell lines compared to either monotherapy. Essentially, the strongest synergistic interactions occurred at high and moderate DMF concentrations of 62.5–125 μg/mL combined with NDV MOIs of 3–5. These conditions consistently produced the lowest cell viability values, indicating a synergistic effect. Nonetheless, this synergy was not restricted to breast cancer cells; normal REF cells showed similar synergistic enhancement, demonstrating that high DMF doses reduce NDV’s natural selectivity toward malignant cells.
Cell viability decreases under most treatment conditions when DMF and NDV are used to fight AMJ- 13 (Figure 4) higher DMF concentrations (125 μg/ml and 62.5 μg/ml) typically exhibit a more potent cytotoxic effect.
Figure 4. Combined Effect of DMF + NDV in AMJ-13 Cells. AMJ-13 cells were treated with three doses of DMF (62.5, 31.25, 15.6 µg/mL) combined with NDV (MOI 1, 3, 5). The strongest reduction in cell viability was observed at DMF 62.5–125 µg/mL with NDV MOI 3–5, indicating potent synergy. Data represent mean ± SEM (n = 3); *P < 0.05; **P < 0.01; ***P < 0.001 compared to monotherapies .
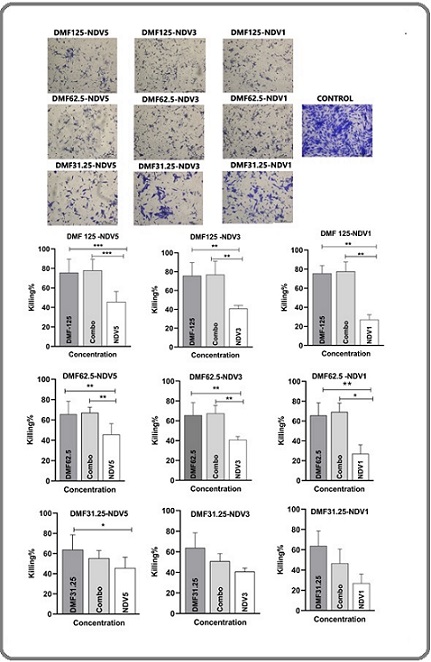
The cytotoxic effects of combinations like (DMF 125 - NDV5, DMF 125 - NDV3, and DMF 62.5 - NDV3) are highly significant (p < 0.01, p < 0.001). The cytotoxic effect appears to be less noticeable at lower concentrations (DMF 31.25 - NDV1).When DMF and NDV are used together, cell viability is typically lower than when either treatment is used alone. The most significant cytotoxic effect that is shown when DMF125 μg/ml and NDV5 are combined suggests a possible synergistic interaction.
Same concentrations of NDV and DMF were used to assess their cytotoxic effects on MCF-7 cells. NDV showed a cytotoxic effect, DMF alone treatment maintained a higher percentage of viable cells, as illustrated (Figure 5).
Figure 5. Combined Effect of DMF + NDV in MCF-7 Cells. MCF-7 cells showed significantly reduced viability when NDV was added to DMF, particularly at DMF 62.5 µg/mL with NDV MOI 5. This supports a synergistic interaction that exceeds the effect of either agent alone. Data represent mean ± SEM (n = 3);*P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001.
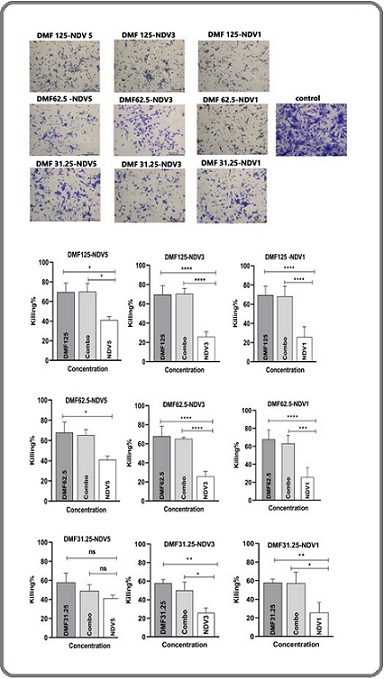
Cell viability was reduced more when DMF and NDV were combined than when DMF was used alone, suggesting a possible synergistic interaction. When combined with DMF, higher concentrations of NDV5 significantly reduced cell viability this effect was statistically significant for all treatment conditions (p < 0.05, *p < 0.01, **p < 0.001). The addition of NDV considerably decreased cell viability even at lower DMF concentrations (31.25 µg/ml), though the effect was not as strong as it was at higher DMF doses. These results support NDV potential as a combinatorial therapeutic approach by indicating that it increases DMF-induced cytotoxicity in MCF-7 cells.
Figure 6 demonstrates the survival of cells treated with different DMF-NDV concentrations.
Figure 6. Combined Effect of DMF + NDV in Normal REF Cells. REF cells exhibited strong synergy at DMF 62.5–125 µg/mL with NDV MOI 3–5, revealing that DMF eliminates the cancer selectivity of NDV and sensitizes normal cells. Data represent mean ± SEM (n = 3); *P < 0.05; **P < 0.01; ***P < 0.001.
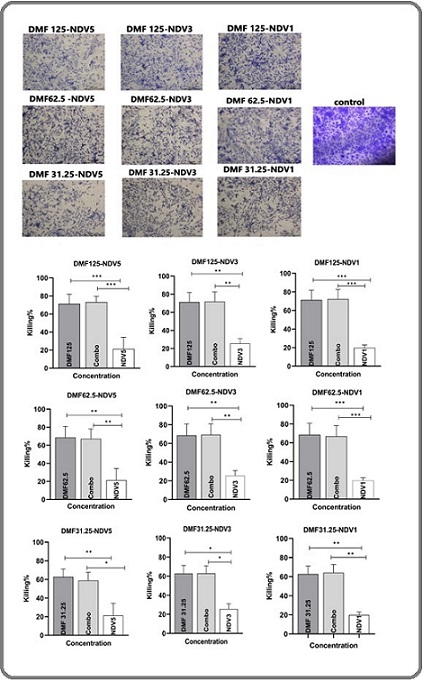
The findings show that NDV alone causes cytotoxicity and that DMF alone has a moderately inhibitory effect on cell viability. However, when compared to either treatment alone, the combination of DMF and NDV dramatically increases cell death. Combining NDV5, NDV3, and NDV1 with 125 µg/ml of DMF significantly reduced cell viability compared to DMF alone (p < 0.001) or NDV alone (p < 0.001). At 62.5 µg/ml, the combination with NDV significantly reduced cell viability (p < 0.01 to p < 0.001). Although the loss in cell viability was less severe at the lowest dose of DMF, the combination treatment still significantly reduced cell survival as compared to monotherapies (p < 0.05 to p < 0.01). collectivly, these results show that while NDV conserves selective toxicity against cancer cells as a monotherapy, the addition of DMF equalizes susceptibility across cancerous and non-cancerous cells. This shift in behavior is dose-dependent and most pronounced at higher DMF concentrations, which correspond to the strongest synergistic CI values.
Chou-Talalay analysis of the cytotoxic effects of dimethyl fumarate and oncolytic NDV combination: We studied the combined cytotoxicity ratios for DMF (125, 62.5, and 31.25 μg/ml) and NDV (1, 3, and 5 MOIs) against AMJ13, MCF-7 and REF cell lines via the CompuSyn isobologram (Tables 1, 2 and 3).
| DMF (µg/ml) | NDV (MOI) | Effect (Fa) | CI Value | Interaction Type |
| 125 | 1 | 0.76 | 0.42748 | synergism |
| 125 | 3 | 0.77 | 0.36858 | synergism |
| 125 | 5 | 0.77 | 0.36864 | synergism |
| 62.5 | 1 | 0.68 | 0.62068 | synergism |
| 62.5 | 3 | 0.67 | 0.70555 | synergism |
| 62.5 | 5 | 0.69 | 0.55197 | synergism |
| 31.25 | 1 | 0.5 | 3.21744 | Antagonism |
| 31.25 | 3 | 0.51 | 4.00482 | Antagonism |
| 31.25 | 5 | 0.5 | 6.84019 | Antagonism |
CI was measured via CompuSyn software
| DMF (µg/ml) | NDV (MOI) | Effect | CI | Interpretation |
| 125 | 1 | 0.69 | 0.38245 | synergism |
| 125 | 3 | 0.7 | 0.28583 | synergism |
| 125 | 5 | 0.7 | 0.28624 | synergism |
| 62.5 | 1 | 0.63 | 0.89 | synergism |
| 62.5 | 3 | 0.65 | 0.59323 | synergism |
| 62.5 | 5 | 0.65 | 0.59731 | synergism |
| 31.25 | 1 | 0.57 | 2.43522 | Antagonism |
| 31.25 | 3 | 0.5 | 16.7524 | Antagonism |
| 31.25 | 5 | 0.5 | 18.8363 | Antagonism |
CI was measured via CompuSyn software
| DMF (µg/ml) | NDV (MOI) | Effect | CI | Interpretation |
| 125 | 1 | 0.69 | 0.56293 | synergism |
| 125 | 3 | 0.7 | 0.44925 | synergism |
| 125 | 5 | 0.69 | 0.56301 | synergism |
| 62.5 | 1 | 0.66 | 0.54158 | synergism |
| 62.5 | 3 | 0.69 | 0.28152 | synergism |
| 62.5 | 5 | 0.67 | 0.43721 | synergism |
| 31.25 | 1 | 0.64 | 0.41266 | synergism |
| 31.25 | 3 | 0.62 | 0.62551 | synergism |
| 31.25 | 5 | 0.58 | 1.44427 | Antagonism |
CI was measured via CompuSyn software
According to synergism (CI < 1), this table demonstrates that higher DMF doses (125, 62.5 µg/ml) efficiently increase NDV-mediated cancer cell killing. Antagonism is evident at lower DMF concentrations (31.25 µg/ml) (CI > 1). The best combination is DMF (125 µg/ml) + NDV (MOI 3-5), which produces the strongest synergy (CI = 0.36–0.37) and the highest effect (Fa = 0.77). The therapeutic potential may be diminished by lower combinations (31.25 µg/ml DMF) that encourage inefficient cell death.
According to this dataset, DMF (125 µg/ml) + NDV (MOI 3-5) exhibits the best synergy (CI = 0.28), which translates to the greatest amount of cancer cell death. However, the synergy between DMF (62.5 µg/ml) and NDV is marginally weaker (CI = 0.59-0.89). The oncolytic action of NDV is not supported by low DMF, as seen by the poor efficacy at low DMF dosages (31.25 µg/ml): Antagonism (CI > 2-18).
Data observed synergism is observed across most doses of DMF + NDV (CI < 1), indicating effective cancer cell killing. Antagonism is observed at the lowest Effect (31.25 µg/ml DMF + 5 MOI NDV), with CI = 1.44 suggesting reduced efficacy.
Discussion
The goal of this investigation was to see if we could boost the effect of virotherapy of Newcastle disease virus against breast cancer cells, hence increasing oncolytic activity in breast cancer, by using the anti-inflammatory drug DMF. Dimethyl fumarate (DMF) has emerged as a viable treatment for a variety of inflammatory and autoimmune conditions. Recent research has investigated its potential as an anticancer agent, particularly in breast cancer [17]. DMF’s major strategy is to block the NF-κB pathway. NF-κB is a transcription factor that affects inflammation and cancer. DMF inhibits NF-κB activity, reducing tumor cell proliferation, invasion, and metastasis [18]. While oncolytic virotherapy employing Newcastle disease virus is a promising method that has good selectivity, tolerability, and activity against breast cancer cells [19]. Furthermore, broad-spectrum anticancer effect, such as anti-lymphoma activity, was demonstrated by the use of Newcastle disease virus. Studies suggest that the combination of oncolytic NDV and standard chemotherapeutic drugs improves cytotoxicity, demonstrating that NDV effectively functions in combination therapy, resulting in synergistic anticancer action [15, 20]. Breast cancer is a malignant disease that starts in the breast tissue. It develops when aberrant breast cells grow uncontrollably, resulting in a tumor. These tumors can then infect surrounding tissues and spread to other sections of the body [21]. According to Chou-Talalay research, the combination of NDV and DMF therapy improved the lethal effect compared to NDV alone. However, the mechanism underlying synergistic activity is unclear. In addition to the possibility that DMF may promote viral replication, the synergistic impact could be attributable to greater anticancer efficacy through combination action, which can lead to higher cell sensitivity to DMF or virotherapy. The activity of this combination therapy may increase the action of each individual treatment, since DMF alone and in combination with NDV is more effective against breast cancer and can induce apoptosis in cancer cells. Recent research has revealed additional pharmacological effects of DMF in a variety of cancer types [22]. In colon cancer models, DMF was demonstrated to inactivate the NRF2 pathway, activate the MAPK signaling pathway, and induce autophagy, leading to oxidative stress and increased cytotoxicity both in vitro and in vivo [23]. It was recently shown that DMF and its metabolite monomethyl fumarate (MMF) alter the GAPDH active site and reduce its enzyme activity, which in turn suppresses aerobic glycolysis and alters cellular immunological functions [24]. Cancer cells predominantly rely on glycolysis for energy production. NDV has been shown to suppress the glycolysis pathway, leading to cancer cell death. Similarly, DMF can disrupt cellular metabolism by depleting glutathione levels, there by inducing oxidative stress. The concurrent disruption of metabolic pathways by both agents could result in enhanced cancer cell cytotoxicity [25]. As a monotherapy, DMF is noticeably more effective. When DMF and NDV are combined, the cytotoxic effect approximates what NDV can accomplish on its own. In addition, DMF induces cell cycle arrest and apoptosis in cervical cancer and melanoma. NF-κB is a key downstream target of DMF in breast cancer [23, 26]. We prove that DMF has anti-tumor action in breast cancer. The research we conducted confirms DMF’s effectiveness as an NF-κB inhibitor and anti-tumor drug, with therapeutic implications. Understanding DMF’s mechanism of action paves the way for its clinical trial to treat aggressive breast cancer [18]. In this study, we concluded that dimethyl fumarate has a toxic effect on normal cells, which is consistent with a study conducted by Wan et al. in 2024 showing that DMF causes heart developmental damage in zebrafish, possibly through the suppression of oxidative stress reactions. This work provides a foundation for future research on the developmental toxicity of DMF to the heart [27, 28].
The results of NDV monotherapy showing preserved selective cytotoxicity toward breast cancer cells, whereas the NDV–DMF combination induced strong synergistic killing in both cancer and normal REF cells, suggests a vital shift in the biological response of normal cells following DMF exposure. Several mechanistic explanations may account for this loss of selectivity.
First, DMF is known to disrupt intracellular redox homeostasis through glutathione (GSH) depletion, covalent modification of cysteine-containing proteins, and induction of oxidative stress [29]. Cancer cells do often possess high basal oxidative stress and rely heavily on glycolytic metabolism and antioxidant buffering systems [30]; therefore, additional oxidative pressure from NDV tends to preferentially kill malignant cells [31]. However, when high DMF doses depletes GSH in normal fibroblasts, their antioxidant capacity becomes significantly compromised [32]. Under these conditions, even moderate NDV-induced reactive oxygen species (ROS) become highly cytotoxic. This redox imbalance effectively removes the natural advantage that normal cells usually possess in resisting NDV-mediated stress. Second, DMF suppresses NF-κB activation, a key prosurvival signaling pathway that regulates inflammatory responses, cytokine production, mitochondrial stability, and expression of anti-apoptotic genes such as Bcl-2 and XIAP [33]. While many cancer cells depend chronically on NF-κB signaling, normal fibroblasts also activate NF-κB in response to viral infection as part of their antiviral defense [34]. By inhibiting this pathway, DMF may reduce the innate antiviral response in REF cells, thereby increasing their permissiveness to NDV replication or NDV-induced stress responses. The suppression of NF-κB can also impair the ability of normal cells to counteract viral-triggered apoptosis, effectively sensitizing them to NDV-induced cytotoxicity. Third, NDV infection itself disrupts cancer cell metabolism, particularly glycolysis, leading to energetic collapse, ferroptosis, and immunogenic cell death [6, 19]. DMF likewise targets glycolytic enzymes such as GAPDH, further impairing energy production [24]. These combined metabolic stresses produce strong synergistic effects in cancer cells. However, the same dual blockade of metabolic pathways in normal cells, after their antioxidant capacity has already been weakened by DMF; may force them beyond a critical survival threshold, explaining the synergistic cytotoxicity observed in REF cells. Therefore, these mechanisms indicate that high DMF doses transforms the intracellular environment of normal fibroblasts into one that closely resembles the stressed metabolic state of cancer cells, making them susceptible to NDV-mediated cell death. This provides a strong mechanistic rationale for the loss of selectivity observed in our study and emphasizes a key limitation of the combination strategy at medium–high DMF concentrations.
In conclusion, the Chou-Talalay analysis demonstrates that combining DMF (an NFκB inhibitor) with oncolytic NDV virotherapy exhibits strong synergism against AMJ13 and MCF-7 cancer cell lines at higher DMF concentrations (125 µg/ml and 62.5 µg/ml), with optimal synergy at DMF (125 µg/ml) + NDV (MOI 3-5) (CI = 0.28-0.37). However, lower DMF doses (31.25 µg/ml) result in antagonism (CI > 1), reducing efficacy. Notably, REF normal cells also show synergism at most doses, indicating cytotoxicity to normal cells, except at the lowest dose (31.25 µg/ml) with NDV (5 MOI), where antagonism is observed (CI = 1.44). for that reason, we recommend minimizing cytotoxicity to normal cells while maintaining efficacy against cancer cells, future studies should focus on reducing DMF concentrations below 62.5 µg/ml, testing intermediate doses (e.g., 40-50 µg/ ml) to identify a threshold that sustains cancer cell killing synergy (CI < 1) while reducing synergism in normal cells (aiming for CI > 1). Additionally, exploring alternative NFκB inhibitors with lower toxicity profiles could further optimize this combination therapy.
Acknowledgments
Sincere thanks to the Experimental Therapy Department, Iraqi Center for Cancer and Medical Genetics Research, Mustansiriyah University, Baghdad, Iraq for the support during the work.
Funding statement
There is no funding source.
Approval
The work approved by scientific committee of Iraqi Center for Cancer and Medical Genetics Research, Mustansiriyah University.
Conflict of interest
There is no competing interest from the author and co-authors.
Author contribution
All authors contributed equally to the work.
References
- The Application of Newcastle Disease Virus (NDV): Vaccine Vectors and Tumor Therapy Yang H, Tian J, Zhao J, Zhao Y, Zhang G. Viruses.2024;16(6). CrossRef
- Pathologic Mechanisms of the Newcastle Disease Virus Zhang D, Ding Z, Xu X. Viruses.2023;15(4). CrossRef
- Advantages and challenges of Newcastle disease virus as a vector for respiratory mucosal vaccines Swart RL , Belov GA . Current Opinion in Virology.2023;62. CrossRef
- Newcastle disease virus: current status and our understanding Ganar K, Das M, Sinha S, Kumar S. Virus Research.2014;184. CrossRef
- Newcastle Disease Virus at the Forefront of Cancer Immunotherapy Burman B, Pesci G, Zamarin D. Cancers.2020;12(12). CrossRef
- Newcastle-disease-virus-induced ferroptosis through nutrient deprivation and ferritinophagy in tumor cells Kan X, Yin Y, Song C, Tan L, Qiu X, Liao Y, Liu W, et al . iScience.2021;24(8). CrossRef
- Hexokinase inhibition using D-Mannoheptulose enhances oncolytic newcastle disease virus-mediated killing of breast cancer cells Al-Ziaydi AG , Al-Shammari AM , Hamzah MI , Kadhim HS , Jabir MS . Cancer Cell International.2020;20. CrossRef
- Papaverine Enhances the Oncolytic Effects of Newcastle Disease Virus on Breast Cancer In Vitro and In Vivo Akram S, Al-Shammari AM , Sahib HB , Jabir MS . International Journal of Microbiology.2023;2023. CrossRef
- The Effect of NF-κB Deactivation on Cancer Cell Response to ALA Mediated Photodynamic Therapy Al-Khafaji ASK , Salman MI , Hassan HA , Al-Shammari AM . Asian Pacific journal of cancer prevention: APJCP.2024;25(6). CrossRef
- Photodynamic therapy for cancer and activation of immune response. in Biophotonics and Immune Responses V. 2010. SPIE Mroz P, Huang YY , Hamblin MR . .
- Combination approaches to potentiate immune response after photodynamic therapy for cancer St Denis TG , Aziz K, Waheed AA , Huang Y, Sharma SK , Mroz P, Hamblin MR . Photochemical & Photobiological Sciences: Official Journal of the European Photochemistry Association and the European Society for Photobiology.2011;10(5). CrossRef
- New Perspectives about Drug Candidates Targeting HTLV-1 and Related Diseases Silva MCM , Pereira RSB , Araujo ACA , Filho EGDS , Dias ADL , Cavalcante KS , Sousa MS . Pharmaceuticals.2023;16(11). CrossRef
- Insight into the mechanism of action of dimethyl fumarate in multiple sclerosis Yadav SK , Soin D, Ito K, Dhib-Jalbut S. Journal of Molecular Medicine.2019;97(4). CrossRef
- The NRF2 pathway as potential biomarker for dimethyl fumarate treatment in multiple sclerosis Hammer A, Waschbisch A, Kuhbandner K, Bayas A, Lee D, Duscha A, Haghikia A, Gold R, Linker RA . Annals of Clinical and Translational Neurology.2018;5(6). CrossRef
- Newcastle disease virus, rituximab, and doxorubicin combination as anti-hematological malignancy therapy Al-Shammari AM , Rameez H, Al-Taee MF . Oncolytic Virotherapy.2016;5. CrossRef
- Drug combination studies and their synergy quantification using the Chou-Talalay method Chou T. Cancer Research.2010;70(2). CrossRef
- DMF Activates NRF2 to Inhibit the Pro-Invasion Ability of TAMs in Breast Cancer Li Y, Jia Y, Xu Y, Li K. Frontiers in Oncology.2021;11. CrossRef
- Dimethyl Fumarate Inhibits the Nuclear Factor κB Pathway in Breast Cancer Cells by Covalent Modification of p65 Protein Kastrati I, Siklos MI , Calderon-Gierszal EL , El-Shennawy L, Georgieva G, Thayer EN , Thatcher GRJ , Frasor J. The Journal of Biological Chemistry.2016;291(7). CrossRef
- Newcastle disease virus suppress glycolysis pathway and induce breast cancer cells death Al-Ziaydi AG , Al-Shammari AM , Hamzah MI , Kadhim HS , Jabir MS . Virusdisease.2020;31(3). CrossRef
- Newcastle disease virus LaSota strain kills human pancreatic cancer cells in vitro with high selectivity Walter RJ , Attar BM , Rafiq A, Tejaswi S, Delimata M. JOP: Journal of the pancreas.2012;13(1).
- A review on methods for diagnosis of breast cancer cells and tissues He Z, Chen Z, Tan M, Elingarami S, Liu Y, Li T, Deng Y, et al . Cell Proliferation.2020;53(7). CrossRef
- Dimethyl Fumarate Attenuates Oxaliplatin-Induced Peripheral Neuropathy without Affecting the Anti-tumor Activity of Oxaliplatin in Rodents Miyagi A, Kawashiri T, Shimizu S, Shigematsu N, Kobayashi D, Shimazoe T. Biological & Pharmaceutical Bulletin.2019;42(4). CrossRef
- Dimethyl fumarate is highly cytotoxic in KRAS mutated cancer cells but spares non-tumorigenic cells Bennett Saidu NE , Bretagne M, Mansuet AL , Just P, Leroy K, Cerles O, Chouzenoux S, et al . Oncotarget.2018;9(10). CrossRef
- Dimethyl fumarate targets GAPDH and aerobic glycolysis to modulate immunity Kornberg MD , Bhargava P, Kim PM , Putluri V, Snowman AM , Putluri N, Calabresi PA , Snyder SH . Science.2018;360(6387). CrossRef
- Oncolytic Newcastle Disease Virus and Photodynamic Therapy as Dual Approach for Breast Cancer Treatment Al-Shammari SMH , Al-Khafaji ASK , Hassan HA , Salman MI , Al-Shammari AM . Asian Pacific journal of cancer prevention: APJCP.2024;25(9). CrossRef
- Dimethylfumarate induces cell cycle arrest and apoptosis via regulating intracellular redox systems in HeLa cells Han G, Zhou Q. In Vitro Cellular & Developmental Biology. Animal.2016;52(10). CrossRef
- Dimethyl fumarate induces cardiac developmental toxicity in zebrafish via down-regulation of oxidative stress Wan M, Liu J, Yang D, Xiao Z, Li X, Liu J, Huang L, et al . Toxicology.2024;503. CrossRef
- Developmental impacts of Nrf2 activation by dimethyl fumarate (DMF) in the developing zebrafish (Danio rerio) embryo Marques ES , Severance EG , Min B, Arsenault P, Conlin SM , Timme-Laragy AR . Free Radical Biology & Medicine.2023;194. CrossRef
- Oxidative stress and pancreatic cancer: a dual role in tumorigenesis and drug toxicity Limbu KR , Chhetri RB , Oh YS , Oak M, Beak DJ , Park E. Toxicological Research.2025;41(6). CrossRef
- Metabolic alterations in cancer cells and therapeutic implications Hammoudi N, Ahmed KBR , Garcia-Prieto C, Huang P. Chinese Journal of Cancer.2011;30(8). CrossRef
- Newcastle disease virus infection induces parthanatos in tumor cells via calcium waves Qu Y, Wang S, Jiang H, Liao Y, Qiu X, Tan L, Song C, et al . PLoS pathogens.2024;20(12). CrossRef
- A cellular model for Friedreich Ataxia reveals small-molecule glutathione peroxidase mimetics as novel treatment strategy Jauslin ML , Wirth T, Meier T, Schoumacher F. Human Molecular Genetics.2002;11(24). CrossRef
- DMF, but not other fumarates, inhibits NF-κB activity in vitro in an Nrf2-independent manner Gillard GO , Collette B, Anderson J, Chao J, Scannevin RH , Huss DJ , Fontenot JD . Journal of Neuroimmunology.2015;283. CrossRef
- NF-kappaB and cancer: how intimate is this relationship Prasad S, Ravindran J, Aggarwal BB . Molecular and Cellular Biochemistry.2010;336(1-2). CrossRef
License

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
Copyright
© Asian Pacific Journal of Cancer Biology , 2026
Author Details