Serum Omentin-1 Level as a Biomarker in Prostate Cancer
Download
Abstract
Objective: To evaluate serum omentin-1 levels in prostate cancer patients relative to healthy controls and to assess its variation between tumor stages and Gleason score categories.
Methodology: This case-control research involved 176 participants (88 prostate cancer patients and 88 controls) from the National Teaching Oncology Hospital in Najaf, Iraq. Serum omentin-1 concentrations were verified by ELISA tests, and relationships with PSA, BMI were measured. ROC analysis was used to assess diagnostic efficacy.
Results: Omentin-1 levels were markedly elevated in prostate cancer patients (43.4 ± 22.6 ng/mL) compared to controls (26.1 ± 6.6 ng/mL, p < 0.01). ROC analysis showed reasonable diagnostic accuracy (AUC = 0.73).
Conclusion: Serum omentin-1 levels are elevated in prostate cancer, and no significant differences were observed across tumor stages or Gleason score categories, indicating that its elevation does not appear to be stage-dependent in this cohort. Further studies are needed to elucidate the mechanistic role of omentin-1 in prostate carcinogenesis, as well as its diagnostic and prognostic potential.
Introduction
Prostate cancer (PCa) is the second most often diagnosed malignancy globally and the fifth leading cause of cancer-related mortality in males, with over 1,460,000 new cases and more than 396,000 deaths recorded in 2022 [1]. From a clinical perspective, PCa exhibits substantial variability; some individuals present with indolent tumors, whilst others progress to aggressive disease characterized by a pronounced propensity for progression and metastasis [2]. In the early stages, symptoms usually appear as lower urinary tract dysfunction, which includes more frequent urination, nocturia, and hesitation. In contrast, severe illness may manifest with urinary retention and bone metastases, mostly impacting the axial skeleton [3]. A full array of risk factors has been definitively established, including advancing age, African ancestry, specific germline mutations, elevated insulin-like growth factor (IGF) levels, and a family history of prostate cancer (PCa). Lifestyle factors, such as diets rich in saturated fats and red meat, tobacco use, alcohol intake, obesity, reduced physical activity, and environmental exposures to chemicals or radiation, may increase the risk of disease [4]. Despite extensive investigation, the etiology of prostate cancer (PCa) remains intricate, including genetic, hormonal, environmental, and inflammatory variables that provoke molecular changes leading to the malignant transformation of prostate epithelial cells [5]. In this context, chronic inflammation, triggered by infections, dietary factors, or hormonal imbalances, plays a substantial role in the etiology of prostate cancer (PCa). Inflammatory mediators induce the generation of reactive oxygen species (ROS), leading to DNA damage and epigenetic alterations. Proliferative inflammatory atrophy (PIA) lesions are considered potential precursors of high-grade prostatic intraepithelial neoplasia (PIN) and cancer [6].
Omentin-1, also termed intelectin-1, is a 34 kDa adipokine encoded by the ITLN1 gene on chromosome 1q21.3. It is the predominant isoform found in human plasma and is secreted mainly from visceral adipose tissue, with markedly higher expression compared to subcutaneous fat [7-11]. Beyond adipose tissue, it is also expressed in endothelial, mesothelial, vascular, and intestinal Paneth cells, exerting autocrine, paracrine, and endocrine functions [12]. Functionally, Omentin-1 modulates insulin and IGF-1 receptor signaling, contributes to glucose and energy metabolism, supports cardiovascular health, and exhibits anti-inflammatory and antioxidant effects [9, 13]. Its circulating levels are regulated by obesity, insulin sensitivity, inflammation, genetic factors, and hormones such as adiponectin, insulin, and fibroblast growth factor-21 [12, 14-16]. In oncology, Omentin-1 displays a context-dependent role. Elevated levels have been reported in mesothelioma, hepatocellular, prostate, colorectal, gastric, and pancreatic cancers, whereas decreased levels occur in renal cell carcinoma [17]. Mechanistically, it can influence tumor progression through apoptosis regulation, Akt pathway modulation, and angiogenic factor secretion, impacting both endothelial and stem cell activity Omentin-1 is regarded as a possible biomarker for cancer progression and prognosis, however its specific role in carcinogenesis is not well-defined. Despite increasing evidence indicating the significance of Omentin-1 in other malignancies, there is a paucity of data concerning its serum levels in prostate cancer patients and their potential correlation with clinical features [18]. The purpose of this research is to investigate the possibility of Omentin-1 as a biomarker for the development and prognosis of prostate cancer by measuring blood levels of the protein and characterizing their association with clinical variables.
Materials and Methods
Study Design
This case-control research was conducted at the National Teaching Oncology Hospital in Najaf, Iraq, from December 2024 to July 2025. This study included two groups: newly diagnosed prostate cancer patients and age-matched healthy controls. The Scientific Committee of the College of Medicine at the University of Basrah granted ethical clearance. Additionally, the hospital ethics board approved this study. All participants provided written informed consent after obtaining detailed information about the study’s aims and methodologies.
There were eighty-eight participants, ranging in age from forty to eighty-five. Recently discovered prostate cancer that was confirmed by histopathology was detected in all patients. Inclusion was determined by having a PSA level higher than 4 ng/mL and not having had any kind of treatment for prostate cancer before, such as hormonal therapy, radiation, or chemotherapy. Meanwhile, 88 age-matched volunteers were included in the control group. Neither their medical history nor their clinical examinations have shown any signs of prostate cancer or benign prostatic hyperplasia (BPH).
Exclusion Criteria
Participants were excluded if they had any of the following:
1- Receiving curative medication for prostate cancer.
2- Patients with a history of other malignancies or previous prostate surgeries.
3- Acute infectious diseases.
4- Chronic liver and kidney diseases.
Physical and biochemical measurements
Age, smoking status, family history, and clinical data including comorbidities such as hypertension, diabetes mellitus, cardiovascular disease, and renal disease were all recorded from all participants. A person’s body mass index (BMI) was determined by recording their anthropometric data, which included their weight and height. Prostate cancer patients had their Gleason scores and TNM stages documented.
Biochemical Analysis
Measurement of Serum Omentin-1
Following the manufacturer’s instructions, serum omentin-1 concentrations were measured using a commercial ELISA kit (Cloud-Clone Corp, USA).
Other Biochemical Parameters
A fully automated VIDAS system developed by Biomerieux in France, which is based on the ELFA (Enzyme Linked Fluorescent Assay) concept, was used to measure PSA concentrations. The fully automated DRI-CHEM NX600 device (Fujifilm, Japan) was used to measure fasting blood glucose, serum urea, serum creatinine, total cholesterol (TC), triglycerides (TG), high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), and very-low-density lipoprotein cholesterol (VLDL-C).
Statistical Analysis
All experiments were performed in triplicate, and the results were expressed as mean ± standard deviation. Data normality was assessed using the Shapiro–Wilk test, and homogeneity of variance was evaluated using Levene’s test. Based on these assumptions, normally distributed variables such as age and BMI were compared using the independent t-test, while non-normally distributed biochemical parameters including Blood Pressure, serum omentin-1, PSA, fasting blood sugar, cholesterol, LDL, VLDL, triglycerides, HDL, creatinine, and BUN, as well as weight, height and Blood Pressure were analyzed using the Mann–Whitney U test. Categorical variables such as residency, smoking status, and comorbidities were assessed using the Chi-square test. Comparisons across patient subgroups for tumor stage and Gleason score were conducted using the Kruskal–Wallis test. Receiver-operating characteristic (ROC) analysis was employed to evaluate the diagnostic performance of serum omentin-1 and PSA in distinguishing patients from controls. A p-value < 0.05 was considered statistically significant. All analyses were conducted using IBM SPSS Statistics 23.0.
Results
Socio-demographic characteristics among the study groups
The socio-demographic features of prostate cancer Patients (88 participants) and a control group (88 healthy adults) are shown in Table 1.
| Parameters | Subgroup | Healthy Control | Patients | P-Value |
| N=88 | N=88 | |||
| Age | - | 59.7±13.4 | 65.2±9.2 | 0.006 |
| BMI | - | 27.02±3.4 | 25.6±3.7 | 0.009 |
| Blood Pressure | Systolic | 124±11.5 | 127±14.9 | 0.3 |
| Diastolic | 80.3±8.05 | 80.4±8.69 | 0.8 | |
| Residency | Urban | 48 (54.5%) | 50 (56.8%) | 0.7 |
| Rural | 40 (45.5%) | 38 (43.2%) | ||
| Non-smoker | 50 (56.8%) | 52 (59.1%) | 0.8 | |
| Smoking status | Ex-smoker | 8 (9.1%) | 9 (10.2%) | |
| Smoker | 30 (34.1%) | 27 (30.7%) | ||
| DM | 13 (14.7%) | 11(12.5%) | 0.2 | |
| Comorbidities | HTN | 8 (9.1%) | 5 (5.7%) | |
| HTN+DM | 4 (4.5%) | 8 (9.1%) | ||
| None | 63 (71.5%) | 64 (72.7%) |
Significant value: p < 0.05, p < 0.01, p < 0.001; Non-significant value: p ≥ 0.05; Independent t-test / Mann– Whitney U / Chi-square test
The research revealed significant differences in age (P=0.006) and body mass index (BMI) (P=0.009) between prostate cancer patients and healthy controls. In contrast, there were no statistically significant differences between the groups regarding residency (urban vs. rural, P=0.7), smoking status (P=0.8), or comorbidities (P=0.2). Similarly, systolic and diastolic blood pressures did not differ significantly between the two groups (P=0.3 and P=0.8, respectively).
Biochemical parameters of study participants
Table 2, serum omentin-1 levels were significantly higher in prostate cancer patients compared with healthy controls (43.4 ± 22.6 vs. 26.1 ± 6.6 ng/mL; P < 0.01).
| Parameters | Healthy Control | Patients | P-Value |
| N=88 | N=88 | ||
| Serum Omentin-1 (ng/ml) | 26.1±6.6 | 43.4±22.6 | <0.01 |
| Total PSA (ng/ml) | 1.68±0.9 | 32.1±31.1 | <0.01 |
| FBS (mg/dl) | 105.5±18.6 | 106.6±21.5 | 0.7 |
| Cholesterol (mg/dl) | 196.9± 23.6 | 198.1± 33.4 | 0.6 |
| Triglycerides (mg/dl) | 138.1± 40.7 | 134.6± 51.5 | 0.2 |
| HDL (mg/dl) | 49.7± 9.8 | 48.04± 12.3 | 0.2 |
| LDL (mg/dl) | 119.6± 25.8 | 123.14± 35.5 | 0.4 |
| VLDL (mg/dl) | 27.6± 8.1 | 26.9± 10.3 | 0.2 |
| S-creatinine (mg/dl) | 0.86± 0.16 | 0.87± 0.24 | 0.5 |
| BUN (mg/dl) | 35.2± 10.3 | 33.46± 12.46 | 0.1 |
*Significant value: p < 0.05 , p < 0.01 , p < 0.001; * Non-significant value: p ≥ 0.05. * All analyses for these variables were performed using the Mann–Whitney U test.
Likewise, PSA levels showed a marked increase in the patient group (32.1 ± 31.1 vs. 1.68 ± 0.9 ng/mL; P < 0.01). For the remaining biochemical parameters, no statistically significant differences were observed between patients and controls, including fasting blood sugar (P = 0.7), cholesterol (P = 0.6), triglycerides (P = 0.2), HDL (P = 0.2), LDL (P = 0.4), VLDL (P = 0.2), serum creatinine (P = 0.5), and BUN (P = 0.1).
Serum omentin levels in prostate cancer patients, stratified by Gleason score and tumor stage
In Table 3, Serum omentin-1 levels showed no significant variation across tumor stages (T1–T4) or Gleason score categories.
| Group | N (patients) (88) | Category | Serum Omentin (ng/ml) | |
| Mean ± SD | P-Value | |||
| 10 | T1 | 49.5±18.3 | 0.753 | |
| Tumor Stage | 25 | T2 | 41.5±20.1 | |
| 30 | T3 | 43.6±24 | ||
| 23 | T4 | 42.5±24.6 | ||
| Gleason Score | 7 | Well-differentiated (score <7) | 44.8± 21.9 | 0.746 |
| 38 | Moderately differentiated (score =7) | 45.3±23 | ||
| 43 | Poorly differentiated (score >7) | 41.8±23 |
*Kruskal–Wallis test were used for comparisons among tumor stages and Gleason score groups. * Statistically significant at p < 0.05; * Statistically non-significant at p > 0.05
Patients in different stages exhibited closely similar mean values (Kruskal–Wallis p = 0.753), and the same pattern was observed across Gleason grades, where well-, moderately-, and poorly differentiated tumors showed nearly identical concentrations (p = 0.746).
ROC Curve Analysis
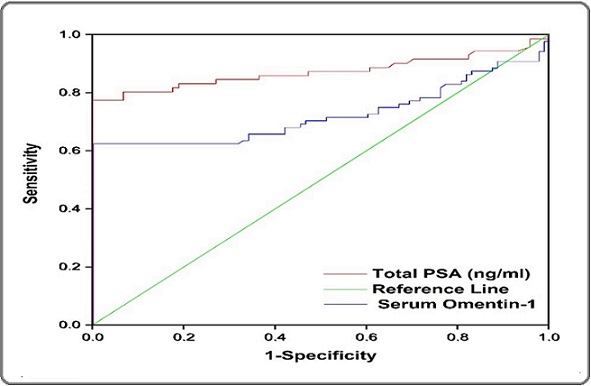
The diagnostic performance of serum Omentin-1 and PSA in distinguishing prostate cancer patients from healthy controls was evaluated using ROC analysis.
Serum Omentin-1: AUC = 0.73, optimal cut-off = 34.6 ng/ml, sensitivity = 63%, specificity = 88%.
PSA: AUC = 0.87, optimal cut-off = 2.04 ng/ml, sensitivity = 85%, specificity = 70%.
These results are summarized in Table 4 and illustrated in Figure 1.
| Markers | AUC | p-value | Cut-off point | Sensitivity | Specificity |
| Serum omentin | 0.73 | <0.0001 | 34.6 | 0.63 | 0.88 |
| PSA | 0.87 | <0.0001 | 2.04 | 0.85 | 0.7 |
Figure 1. ROC Curve Analysis of Serum Omentin (Blue line), and PSA (Red line) in the Study Population.
Discussion
Prostate cancer (PCa) continues to represent a major public health burden, accounting for approximately 19% of all new cancer cases and 9% of cancer-related deaths in men globally [19]. We investigated the clinical significance of serum omentin-1 in PCa patients vs healthy controls in an effort to determine its possible role as a biomarker. Serum levels of omentin-1 were markedly elevated in prostate cancer patients (43.4 ± 22.6 ng/mL) when compared with healthy controls (26.1 ± 6.6 ng/mL, P < 0.01). The very high serum values likely point to its potential involvement in the pathophysiology of prostate cancer disease and its utility as a biomarker. Findings are in line with those of Zhou et al. [7], Uyeturk et al., and Fryczkowski et al., that demonstrated pronounced omentin-1 levels in PCa that could be established as a non-invasive diagnostic marker [20,21]. When serum omentin-1 levels were compared across prostate cancer stages (T1–T4), no statistically significant differences were observed among the groups. Although patients in stage T1 showed numerically higher mean concentrations compared with more advanced stages, these variations did not reach statistical significance, indicating that the observed pattern may simply reflect natural biological variability rather than a stage-dependent effect. Accordingly, the current data do not support any definitive interpretation regarding omentin-1 behavior across tumor stages. Similarly, when stratified by Gleason score, omentin-1 levels did not differ significantly between well-differentiated, moderately differentiated, and poorly differentiated tumors. These findings align with those reported by Uyeturk et al. [20], who also found no association between circulating omentin-1 and histopathological grade, suggesting that omentin-1 is unlikely to reflect tumor aggressiveness. Evidence from other malignancies further supports the context-dependent and variable behavior of omentin-1. Shen et al. [22], in renal cancer and Fazeli et al. [23], in colorectal cancer both reported inconsistent relationships between omentin-1 and tumor stage, reinforcing that this adipokine may not follow a uniform pattern across different cancers. Experimental data offer several mechanistic hypotheses but do not establish causality in humans. Mogal et al. [24], reported that omentin suppresses prostate cell proliferation by downregulating the tumor suppressor gene Nkx3.1, while Cheng et al. [25], demonstrated that mesenchymal stem cells in the tumor microenvironment drive androgen-dependent PCa cells toward androgen-independent, more aggressive forms. This supports the concept that decreased omentin-1 levels in advanced stages may reflect the loss of protective metabolic regulation.
Apart from prostate carcinoma, a number of researchers have investigated the role of omentin in other cancers. Omentin inhibited hepatocellular carcinoma formation by activation of the JNK pathway and by upregulating p53 [26], and Ji et al. [27], observed enhanced apoptosis in colon cancer stem cells. Yet contrary evidence does exist: Dec et al. [18], demonstrated that omentin promoted colon cancer growth, and Zhang et al. [28], shows that colorectal carcinoma cells secrete omentin locally, which may accelerate tumorigenesis through glucose uptake [29] and PI3K/Akt signaling [30], from the well-established pathway that promotes prostate cancer [31]. Omentin-1 is primarily synthesized by visceral adipose tissue, where the expression of PI3K/Akt contributes to mesenchymal stem cell survival [18]. Moreover, other components of the tumor microenvironment (adipocytes, macrophages and endothelial cells) release proangiogenic mediators (VEGF and fibroblast growth factors) that promote tumor invasion and progression [32, 33].
We obtained findings based on the ROC analysis which indicate that serum omentin-1 had a moderate discriminative performance, with an AUC of 0.73, sensitivity of 63%, and specificity of 88%, corresponding to an optimal cut-off value of 34.6 ng/mL. In contrast, PSA showed superior diagnostic accuracy, achieving an AUC of 0.87 with a cut-off value of 2.04 ng/mL (sensitivity 85%, specificity 70%). These results indicate that although PSA remains the more robust marker, omentin-1 may still provide additional diagnostic value as a complementary biomarker, This observation agrees with Fryczkowski et al.’s findings [21] on omentin’s diagnostic potential as an adjunct biomarker to PSA.
Molecularly, prior studies imply that omentin can modulate several pathways. Zhang et al. [26], showed that omentin suppresses hepatocellular carcinoma growth by the upregulation of p21 and p53 proteins, induces apoptosis by promoting Bax/Bcl-2 transcription, and inhibits caspase-3 activation. Omentin also was found to activate the Akt pathway and modulate PI3K/Akt-eNOS-Ras signaling that could be implicated, and promote or inhibit cancer depending on cellular context [18, 27-34]. While these pathways enable theoretically a rationale for omentin-1’s potential role in cancer biology, the current study reveals no direct mechanistic relationships. Our data show serum omentin-1 is high in PCa and different between diseases; however, these observations indicate associations and not causality. Such mechanistic studies along with longitudinal designs are needed to better elucidate whether omentin-1 exerts a causal role in PCa progression or represents the only metabolic perturbations of the disease. The present case–control study is limited in some aspects as follows:
First: The design enables comparisons between prostate cancer patient’s vs healthy controls, yet does not allow for causal inference in terms of relation of serum omentin-1 levels to the development/progression of prostate cancer. Second: The trial was performed in only one city, and the sample had a small size, so the results may not be applicable over larger populations. Third: Dietary habits, frequency of physical activity and unmeasured metabolic or hormonal parameters were not fully controlled and could have affected omentin-1 levels.
Finally, the study had no longitudinal follow-up to assess temporal alteration of omentin-1 at different stages of disease onset and after different interventions. Nevertheless, multicenter prospective studies are required to confirm these observations and to better clarify the potential clinical relevance of omentin-1 in PCa.
In conclusion, serum omentin-1 was significantly elevated in prostate cancer patients compared to healthy controls, suggesting a possible association between elevated omentin-1 levels and the presence of prostate malignancy. No significant differences were observed with respect to prostate cancer stage and Gleason score levels in this cohort, however, indicating that omentin-1 does not reflect disease stage or histological aggressiveness. Overall, the findings imply omentin-1 could be used as an adjunct biomarker in prostate cancer detection. However, additional work is necessary to elucidate its biological function and to evaluate a reasonable diagnostic or prognostic potential in clinical practice.
Acknowledgments
This research is supported by the Department of Biochemistry, College of Medicine, University of Basrah, as part of the Master graduation requirements.
Funding
This research did not receive any external funding. The study was conducted using departmental resources of the Department of Biochemistry, College of Medicine, University of Basrah.
Author contributions
Conceptualization: AMM and AIA; Methodology: AMM and AIA; Software: AF; Validation: HJS; Formal Analysis: AMM and AIA; Investigation: AMM; Supervision, Project Administration, and Data Curation: AIA and HJS; Writing – Original Draft Preparation: AMM, AIA. Writing – Review & Editing: AMM and AIA. All authors have read and approved the final version of the manuscript
Conflicts of interest
The authors deny there are any financial conflicts of interest.
Data availability upon reasonable request.
References
- Recent Patterns and Trends in Global Prostate Cancer Incidence and Mortality: An Update Schafer E.J., Laversanne M., Sung H., Soerjomataram I., Briganti A., Dahut W., Bray F., Jemal A.. Eur Urol.2025;Mar;87(3):302-313. CrossRef
- Cellular and Molecular Mechanisms Underlying Prostate Cancer Development: Therapeutic Implications Testa U., Castelli G., Pelosi E.. Medicines (Basel.2019;30;6(3):82. CrossRef
- Epidemiology of Prostate Cancer Rawla P.. World J Oncol.2019;Apr;10(2):63-89. CrossRef
- Underlying Features of Prostate Cancer-Statistics, Risk Factors, and Emerging Methods for Its Diagnosis Berenguer C.V., Pereira F., Câmara J.S., Pereira J.A.M.. Curr Oncol.2023;Feb 15;30(2):2300-2321. CrossRef
- Deep Learning-Based Multi-Omics Integration Robustly Predicts Relapse in Prostate Cancer Wei Z., Han D., Zhang C., Wang S., Liu J., Chao F., Song Z., Chen G.. Front Oncol.2022. CrossRef
- The inflammatory microenvironment and microbiome in prostate cancer development Sfanos K.S., Yegnasubramanian S., Nelson W.G., Marzo A.M.. Nat Rev Urol.2018;Jan;15(1):11-24. CrossRef
- Altered circulating levels of adipokine omentin-1 in patients with prostate cancer Zhou L., He W., Wang W., Zhou D.. Onco Targets Ther.2019. CrossRef
- Clinical significance of circulating omentin levels in various malignant tumors: Evidence from a systematic review and meta-analysis Arjmand M.H., Moradi A., Akbari A., Mehrad-Majd H.. Cytokine.2020;125(154869). CrossRef
- Plasma level of omentin-1, its expression, and its regulation by gonadotropin-releasing hormone and gonadotropins in porcine anterior pituitary cells Respekta N., Pich K., Mlyczyńska E., Dobrzyń K., Ramé C., Kamiński T., Smolińska N., Dupont J., Rak A.. Sci Rep.2023;(v 7;13(1):19325). CrossRef
- The association between serum omentin level and bodyweight: A systematic review and meta-analysis of observational studies Arab A., Moosavian S.P., Hadi A., Karimi E., Nasirian M.. Clin Nutr ESPEN.2020;39. CrossRef
- Omentin plasma levels and gene expression are decreased in obesity Souza Batista CM , Yang RZ , Lee MJ , Glynn NM , Yu DZ , Pray J , Ndubuizu K , et al . Diabetes.2007;Jun;56(6):1655-61. CrossRef
- Adipose Tissue-Derived Omentin-1 Function and Regulation Watanabe T., Watanabe-Kominato K., Takahashi Y., Kojima M., Watanabe R.. Compr Physiol.2017;Jun 18;7(3):765-781. CrossRef
- Association of Serum Omentin-1 Concentration with the Content of Adipose Tissue and Glucose Tolerance in Subjects with Central Obesity Sperling M., Grzelak T., Pelczyńska M., Bogdański P., Formanowicz D., Czyżewska K.. Biomedicines.2023;24;11(2):331. CrossRef
- New adipokines vaspin and omentin. Circulating levels and gene expression in adipose tissue from morbidly obese women Auguet T., Quintero Y., Riesco D., Morancho B., Terra X., Crescenti A., Broch M., et al . BMC Med Genet.2011;12(60). CrossRef
- Association of a fat-derived plasma protein omentin with carotid artery intima-media thickness in apparently healthy men Shibata R., Takahashi R., Kataoka Y., Ohashi K., Ikeda N., Kihara S., Murohara T., Ouchi N.. Hypertens Res.2011;Dec;34(12):1309-12. CrossRef
- Changes of serum omentin levels and relationship between omentin and adiponectin concentrations in type 2 diabetes mellitus Yan P., Liu D., Long M., Ren Y., Pang J., Li R.. Exp Clin Endocrinol Diabetes.2011;Apr;119(4):257-63. CrossRef
- The Role of Omentin-1 in Cancers Development and Progression Dec P., Poniewierska-Baran A., Modrzejewski A., Pawlik A.. Cancers (Basel).2023;26;15(15):3797. CrossRef
- Omentin-1 effects on mesenchymal stem cells: proliferation, apoptosis, and angiogenesis in vitro Yin L., Huang D., Liu X., Wang Y., Liu J., Liu F., Yu B.. Stem Cell Res Ther.2017;Oct 10;8(1):224. CrossRef
- Cancer statistics, 2018 Siegel RL , Miller KD , Jemal A. CA: a cancer journal for clinicians.2018;68(1). CrossRef
- Serum omentin level in patients with prostate cancer Uyeturk U, Sarıcı H, Kın Tekce B, Eroglu M, Kemahlı E, Uyeturk U, Gucuk A. Medical Oncology.2014;31(4). CrossRef
- Circulating Levels of Omentin, Leptin, VEGF, and HGF and Their Clinical Relevance with PSA Marker in Prostate Cancer Fryczkowski M., Bułdak R. J., Hejmo T., Kukla M., Żwirska-Korczala K.. Disease Markers.2018;2018. CrossRef
- Circulating levels of adipocytokine omentin-1 in patients with renal cell cancer Shen X, Zhang L, Che H, Zhang Y, Yang C, Zhou J, Liang C. Cytokine.2016;77. CrossRef
- Circulating levels of novel adipocytokines in patients with colorectal cancer Fazeli MS , Dashti H, Akbarzadeh S, Assadi M, Aminian A, Keramati MR , Nabipour I. Cytokine.2013;62(1). CrossRef
- Haploinsufficient Prostate Tumor Suppression by Nkx3.1 Mogal AP , Van Der Meer R, Crooke PS , Abdulkadir SA . Journal of Biological Chemistry.2007;282(35). CrossRef
- The role of mesenchymal stem cells in promoting the transformation of androgen-dependent human prostate cancer cells into androgen-independent manner Cheng J, Yang K, Zhang Q, Yu Y, Meng Q, Mo N, et al . Scientific Reports.2016;6(1). CrossRef
- Omentin-1, a new adipokine, promotes apoptosis through regulating Sirt1-dependent p53 deacetylation in hepatocellular carcinoma cells Zhang Y, Zhou L. European Journal of Pharmacology.2013;698(1-3). CrossRef
- The effect of omentin-1 on the proliferation and apoptosis of colon cancer stem cells and the potential mechanism Ji H, Wan L, Zhang Q, Chen M, Zhao X. Journal of B.U.ON.: official journal of the Balkan Union of Oncology.2019;24(1).
- Autocrine action of adipokine omentin‑1 in the SW480 colon cancer cell line Zhang Y, Zhao X, Chen M. Oncology Letters.2019. CrossRef
- Identification of omentin as a novel depot-specific adipokine in human adipose tissue: possible role in modulating insulin action Yang R, Lee M, Hu H, Pray J, Wu H, Hansen BC , Shuldiner AR , et al . American Journal of Physiology. Endocrinology and Metabolism.2006;290(6). CrossRef
- Omentin-1 Stimulates Human Osteoblast Proliferation through PI3K/Akt Signal Pathway Wu S, Liang Q, Liu Y, Cui R, Yuan L, Liao E. International Journal of Endocrinology.2013;2013. CrossRef
- AR Signaling and the PI3K Pathway in Prostate Cancer Crumbaker M, Khoja L, Joshua AM . Cancers.2017;9(4). CrossRef
- Periprostatic adipose tissue and prostate cancer progression: new insights into the tumor microenvironment Toren P, Venkateswaran V. Clinical Genitourinary Cancer.2014;12(1). CrossRef
- Mesenchymal stem cells: key players in cancer progression Ridge SM , Sullivan FJ , Glynn SA . Molecular Cancer.2017;16(1). CrossRef
- Omentin prevents myocardial ischemic injury through AMP-activated protein kinase- and Akt-dependent mechanisms Kataoka Y, Shibata R, Ohashi K, Kambara T, Enomoto T, Uemura Y, Ogura Y, et al . Journal of the American College of Cardiology.2014;63(24). CrossRef
License

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
Copyright
© Asian Pacific Journal of Cancer Biology , 2026
Author Details