Immunohistochemical Study of Tissue Expression of Trophoblast Cell Surface Antigen 2 (TROP2) Protein in Prostatic Carcinoma and Benign Hyperplasia
Download
Abstract
Background: Prostate cancer is the second most common malignancy affecting males. TROP2 is a cell surface marker of embryonic stem cells that has been implicated in development of many cancers.
Objective: This study aimed to investigate the immunohistochemical expression of TROP2 Protein in PCa and BPH tissues and explore TROP2 association with the histopathological parameters of PCa.
Materials and Methods: a cross-sectional analytical study was conducted in the pathology laboratory of Suez Canal University Hospital on thirty PCa and fifteen BPH specimens (n=45). Sample was collected from the archived blocks of BPH and prostatic carcinoma. TROP2 expression was assessed using immunohistochemistry.
Results: TROP2 showed positive membranous staining in prostatic epithelial cells of 100% of PCa & in eleven out of fifteen BPH specimens and the difference between the two groups was statistically significant (p-value >0.001). Moreover, high TROP2 expression was found in nineteen PCa and in only four BPH specimens and the difference between the two groups regarding TROP2 staining intensity was also statistically significant (p-value= 0.005). TROP2 showed positive expression in 100% of specimens showing positive perineural invasion and a statistically significant association was found between both TROP2 staining intensity and perineural invasion (p-value >0.05). In addition, TROP2 showed statistically significant validity in discriminating between PCa and BPH (p-value <0.001).
Conclusions: TROP2 is a sensitive prostatic immunohistochemical marker and its expression in PCa is higher than that in BPH. TROP2 is associated with poor prognostic factors of PCa as perineural invasion.
Introduction
Prostatic carcinoma is an important health problem of elderly males. As the prostate is a hormone dependent secretory gland, it may undergo a group of diseases namely epithelial hyperplasia, infection, inflammation, benign and malignant neoplastic enlargement.
Most prostatic diseases are benign, however, malignant neoplasms are very important to be studied because of the poor prognosis and high mortality associated with metastases. So, it is important to find new diagnostic tools that allow early tumor detection, better treatment chances and better prognosis [1, 2].
Prostate cancer (PCa) is the second most common malignancy in males next to lung cancer and the fifth cause of cancer death in males worldwide according to WHO in 2020. According to Global Cancer Observatory statistics in December 2020, the Middle East reported 51,649 new prostate cancer diagnoses, corresponding to 3.7% of global cases. In Egypt, PCa was found to be the seventh highly detected cancer representing 7.2% of the total diagnosed cancer cases in Egyptian males [3-5].
Trophoblast cell surface antigen 2 (TROP2), is a stem cell marker that shows membranous expression in several normal adult tissues as endocrine, exocrine glands, GIT, heart, kidneys, larynx, salivary gland, skin, thymus, tonsils, trachea, lung, urothelium, prostate, seminal vesicles and female genital tract TROP2 is also a protooncogene whose overexpression is involved in promoting tumor cell growth, proliferation and metastasis [6-8].
TROP2 has been implicated in development and progression of breast and ovarian cancers [9]. TROP2 was found to be important therapeutic target in triple negative breast cancer, advanced lung cancer, and metastatic urothelial carcinoma [6, 10].
Some studies have investigated the expression of TROP2 in prostate cancer and it was found that TROP2 expression was higher in prostate cancer tissue samples compared to normal prostate tissue and that TROP2 expression was associated with poorer overall survival [11-13].
While these studies suggest that TROP2 may play a role in the development and progression of prostate cancer, few studies have investigated the expression of TROP2 in BPH tissue samples [14, 15]. The current study aimed to investigate the immunohistochemical tissue expression of TROP2 proteins in both prostate cancer and BPH tissue samples and explore TROP2 association with the histopathological parameters of PCa to determine if TROP2 protein can be used as potential prognostic biomarkers of prostate cancer. Moreover, this study aimed to give insights into the diagnostic performance of TROP2 expression in differentiating PCa and BPH.
Materials and Methods
The current work is a cross-sectional analytical study, conducted in the pathology laboratory of Suez Canal University Hospital on thirty prostatic carcinoma specimens and fifteen benign prostatic hyperplasia specimens. They were gathered from records and formalin fixed paraffin embedded tissue blocks during the interval between 2013 and 2019.
Hematoxylin & eosin staining
Sections were prepared from the paraffin embedded tissue blocks of PCa and BPH specimens at thickness of 5 µm and were stained with Hematoxylin & eosin (H&E) stains. H&E slides were initially examined for histopathological features according to the 2022 WHO classification of prostatic tumors that are tumor histologic type, Gleason WHO/ISUP grade grouping, and the estimated approximate percentage of prostate involved by tumor in examined slides, presence or absence of lymphovascular and perineural invasion.
Immunohistochemical staining
Other sections were prepared on positive charged slides to be stained with TROP2 primary antibody. Normal skin tissue was used as a positive control [13]. Immunohistochemical staining for TROP2 was conducted using monoclonal anti-TROP2 IgG clone (F-5): sc-376181 purified antibody that was used according to the suggested protocol included within companies’ data sheets (Santa Cruz).
Staining Technique followed the following steps: at first, sections were deparaffinized in xylene for 30 minutes then rehydrated for 20 minutes in descending grades of alcohol followed by distilled water for 6 minutes. Antigen heat induced epitope retrieval, was done by immersing slides in high pH retrieval buffer solution, for 20 minutes at 98oC in microwave. Slides were cooled gradually followed by immersing in distilled water and wash buffer for 6 minutes. Ten minutes of immersion in hydrogen peroxide was performed for blocking endogenous peroxidase activity followed by immersing in wash buffer for 6 minutes. Slides were incubated with primary antibody for 2 hours at room temperature then washed with wash buffer for 6 minutes before applying the secondary antibody for 25 minutes. After washing with wash buffer the slides were incubated with diaminobenzidine chromogen for 10 minutes, then washed with distilled water then counterstained with ready to use Mayer’s hematoxylin for 10 minutes.
Evaluation of immunohistochemical staining of TROP2
PCa and BPH slides stained by TROP2 marker were microscopically examined. The membranous staining of TROP2 in prostatic epithelial cells was quantified based on proportion of stained tumor cells. The membranous expression of TROP2 was scored as negative if percentage of stained tumor cells is less than 10% and as positive if percentage of stained tumor cells is more than 11%. Positive TROP2 expression was subdivided into +1 (low) if percentage of positively stained tumor cells is 11-49% and +2 (high) if percentage of positively stained tumor cells is 50-100% [13].
Data analysis
Descriptive analysis was adopted for percentages and means of different findings. Association between level of TROP2 expression and the different histopathological parameters was calculated using Spearman correlation test and chi square test. Receiver operating characteristic (ROC) curve analysis for TROP2 expression was conducted. Statistical significance was considered at P- value <0.05. Statistical analysis was done using the software SPSS (Version 21) for windows. Visual presentation of results was displayed using tables & figures.
Results
A total of forty five prostatic specimens were included in the present study, of which thirty were PCas (Table 1) (Figure 1) and fifteen were BPH (Figure 2).
| Parameters | n |
| Dominant Growth Pattern | |
| Acinar | 28 |
| Ductal | 2 |
| Positive Cribriform Pattern | 25 |
| Gleason Grade | |
| Grade group 3 | 4 |
| Grade group 4 | 8 |
| Grade group 5 | 18 |
| Gleason Score, Median | 30 |
| Comedo necrosis | |
| None | 13 |
| Focal | 10 |
| Diffuse | 7 |
| Approximate % of prostate involved by tumor | |
| Less than 80% | 10 |
| 80 – 99% | 8 |
| 100% | 12 |
| Lymphocytic infiltration | 12 |
| Positive LVI | 5 |
| Positive PNI | 9 |
LVI, Lymphovascular Invasion. PNI, Perineural Invasion
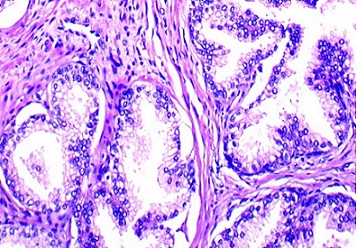
Figure 1. Prostatic Acinar Adenocarcinoma, Gleason grade (4+4), Grade Group 4, H and E Staining, Original Magnification x100.
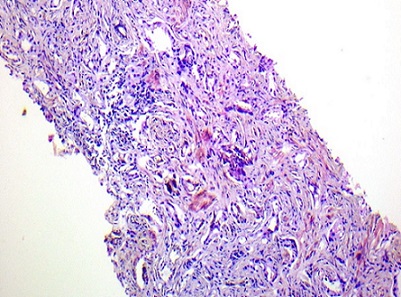
Figure 2. Benign Prostatic Hyperplasia Showing Dilated Prostatic Acini with Double Layered Epithelium, H and E Staining, Original Magnification x400.
TROP2 immunohistochemical staining in benign prostatic hyperplasia and prostatic carcinoma specimens
TROP2 expression showed membranous staining in in prostate basal epithelial cells of 100% (thirty) of PCa (Figure 3 and 4) and in eleven BPH specimens.
Figure 3. Positive TROP2 Membranous Staining Score 2 in Prostatic Acinar Adenocarcinoma, Gleason Grade (4 +4), Grade Group 4, Immunohistochemical Staining, Original Magnification x200.
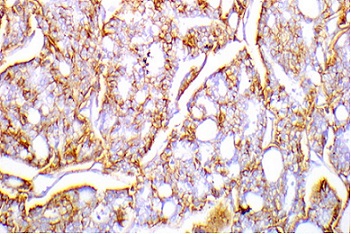
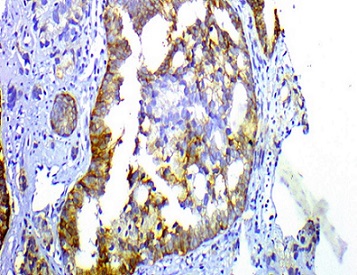
Figure 4. Positive TROP2 Membranous Staining Score 1 in Prostatic Acinar Adenocarcinoma, Grade (4 +3), Grade Group 3, Immunohistochemical Staining, Original Magnification x200.
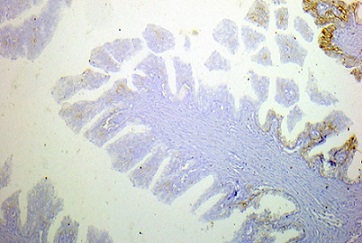
TROP2 expression was negative in four BPH specimens (Figure 5) with statistically significant difference between the two groups (p-value= 0.009) (Table 2).
Figure 5. Negative TROP2 Membranous Staining Score 0 (less than 10%) in Benign Prostatic Hyperplasia, Immunohistochemical Staining, Original Magnification x100.
As regards TROP2 staining intensity, high TROP2 expression was found in nineteen PCa (Figure 3) and in only four BPH specimens and low TROP2 expression was found in seven BPH and in only eleven PCa specimens (Figure 4) and the difference between the two groups concerning staining intensity was statistically significant (p-value= 0.005) (Table 2).
| Parameters | BPH (n = 15) | PC (n = 30) | p-value |
| TROP2 Expression | |||
| Negative | 4 (26.7%) | 0 | 0.009 *F |
| Positive | 11 (73.3%) | 30 (100.0%) | |
| Median percentage of TROP2 positivity | |||
| Median | 20.0 (0.0 – 55.0) | 85.0 (40.0 – 90.0) | <0.001 *M |
| TROP2 Staining Intensity | |||
| No Staining | 4 (26.7%) | 0 | 0.005 *F |
| Low | 7 (46.7%) | 11 (36.7%) | |
| High | 4 (26.7%) | 19 (63.3%) |
BPH: Benign Prostatic Hyperplasia, PCa: Prostatic Carcinoma, TROP2: Trophoblast Cell Surface Antigen 2, *statistically significant p-value at p<0.05, F. Fischer’s exact test, M. Mann Whitney test.
Association between TROP2 immunohistochemical staining and histopathological characteristics in prostatic carcinoma group
Regarding Gleason grade, among nineteen specimens that showed high TROP2 expression, eleven specimens were Gleason grade group 5, five specimens were Gleason grade group 4 and only three specimens were Gleason grade group 3. TROP2 expression was also high in eight out of twelve specimens showing positive lymphocytic infiltration. Low TROP2 expression was found in three out of five specimens revealing positive lymphovascular invasion. However, p-value of association between TROP2 expression and all these studied histopathological parameters of PCa was greater than 0.05. Interestingly, a statistically significant association was found between TROP2 staining intensity and perineural invasion (p-value >0.05) (Table 3).
| Parameters | n | TROP2 expression | ||
| Low (n=11) | High (n=19) | p-value | ||
| Dominant Growth Pattern | ||||
| Acinar | 28 | 10 (90.9%) | 18 (94.7%) | 1.00 F |
| Ductal | 2 | 1 (9.1%) | 1 (5.3%) | |
| Positive Cribriform Pattern | 25 | 10 (90.9%) | 15 (78.9%) | 0.626 F |
| Gleason Grade | ||||
| Grade group 3 | 4 | 1 (9.1%) | 3 (15.8%) | 1.00 F |
| Grade group 4 | 8 | 3 (27.3%) | 5 (26.3%) | |
| Grade group 5 | 18 | 7 (63.6%) | 11 (57.9%) | |
| Gleason Score, Median | 30 | 9.0 (8.0 – 9.0) | 9.0 (8.0 – 9.0) | 0.800 M |
| Comedo necrosis | ||||
| None | 13 | 4 (36.4%) | 9 (47.4%) | 0.622 F |
| Focal | 10 | 5 (45.5%) | 5 (26.3%) | |
| Diffuse | 7 | 2 (18.2%) | 5 (26.3%) | |
| Approximate % of prostate involved by tumor | ||||
| Less than 80% | 10 | 3 (27.3%) | 7 (36.8%) | 0.095 F |
| 80 – 99% | 8 | 1 (9.1%) | 7 (36.8%) | |
| 100% | 12 | 7 (63.6%) | 5 (26.3%) | |
| Lymphocytic infiltration | 12 | 4 (36.4%) | 8 (42.1%) | 1.00 F |
| Positive LVI | 5 | 3 (27.3%) | 2 (10.5%) | 0.327 F |
| Positive PNI | 9 | 6 (54.5%) | 3 (15.8%) | 0.042 * F |
TROP2: Trophoblast Cell Surface Antigen 2, M. Mann Whitney test, F. Fischer’s exact test. LVI: Lymphovascular Invasion. PNI: Perineural Invasion. * Statistically significant p-value at p<0.05.
Receiver operating characteristic (ROC) curve analysis for TROP2 expression
Regarding the diagnostic performance of TROP2 expression in differentiating PCa and BPH, TROP2 showed statistically significant validity in discriminating between PCa and BPH (p-value <0.001) (Table 4, Figure 6).
| AUC | Standard Error | 95% CI | z statistic | p-value (Area=0.5) | |
| TROP2 | 0.902 | 0.0495 | 0.776 to 0.970 | 8.13 | <0.001 * |
AUC: Area under the ROC curve. CI: Confidence interval
Figure 6. ROC Curve of TROP2 Expression Level for Discrimination between PCa and BPH (n = 45).
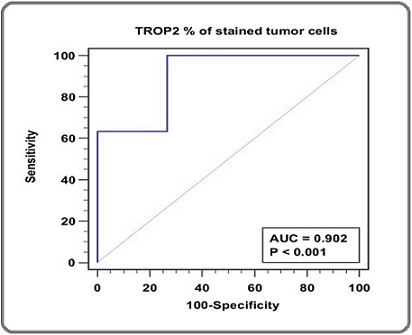
TROP2 showed 100% sensitivity and 73.33% specificity in detecting PCa (Table 5).
| Cut-off value | Sensitivity | Specificity | +LR | -LR | +PV | -PV | |
| 95% CI | 95% CI | 95% CI | 95% CI | 95% CI | 95% CI | ||
| TROP2 | >25 | 100 | 73.33 | 3.8 | 0 | 88.2 | 100 |
| (88.4-100) | (44.9 -92.2) | (1.62 -8.68) | (76.4-94.6) |
CI: Confidence interval. LR: Likelihood ratio. PV: Predictive value
Discussion
The study was performed on thirty prostatic carcinoma specimens and fifteen benign prostatic hyperplasia specimens. TROP2 showed positive membranous expression in all prostatic carcinoma and eleven out of fifteen BPH specimens and the difference between the two groups was statistically significant (p-value >0.001). The findings are largely comparable to those found in other research that studied TROP2 expression in prostatic tumors, in which TROP2 revealed positive membranous expression in prostatic epithelial cells and its expression in PCa cells ranged between 63.4 & 100% &was higher than its expression in benign prostatic epithelial cells (75%) [13-15]. This variation in percentage of expression between studies is mostly related to differences in sample size, type of specimens& primary antibody nature.
Additionally, TROP2 staining differed between PCa and BPH groups in its intensity (p-value =0.005), in which high TROP2 expression was found in nineteen PCa and in only four BPH specimens and low TROP2 expression was found in seven BPH and eleven PCa specimens. Comparable finding was reported in previous study [14] which also documented that high TROP2 intensity was more in PCa (50%) than in BPH specimens (30%) and low TROP2 intensity was also greater in BPH study group (45%).
Our study observed a positive association between TROP2 intensity and Gleason grades, where specimens with higher Gleason grades exhibit stronger TROP2 staining. This correlation highlights TROP2’s role in prostate cancer aggressiveness, supporting its known function in enhancing the expression of proto-oncogenes and promoting self-renewal and proliferation of cancer cells. TROP2 upregulation may thus contribute to the progression and malignant behavior of prostate cancer, with more aggressive cases showing stronger TROP2 expression [8, 14, 16, 17].
However, despite the observed trend, association between TROP2 intensity and Gleason grade wasn’t statistically significant. This finding is in line with several studies, though it contrasts with research by Akarken & Dere (2021), which reported a statistically significant link between TROP2 expression and Gleason grades. Such discrepancies might arise from variations in sample sizes, TROP2 scoring methodologiesor interpretation of staining intensity across studies.
The present work showed positive TROP2 was in all (nine) specimens with positive perineural invasion, with a statistically significant association (p-value >0.05). This result aligns with previous findings by Akarken & Dere (2021) and partially reflects TROP2 involvement in promoting PCa invasiveness. TROP2 is thought to contribute to perineural invasion by facilitating epithelial-mesenchymal transition (EMT) through the upregulation of p-21-activated kinase 4 [8, 17].
Moreover, TROP2 was expressed in all (five) prostatic carcinoma samples that showed positive lymphovascular invasion. Unlike perineural invasion, however, this association was not statistically significant in our study, which diverges from Akarken & Dere’s (2021) findings of a significant association between TROP2 expression and lymphovascular invasion. This discrepancy may stem from the small sample size of specimens with lymphovascular invasion in our research or differences in statistical methods. Nonetheless, the consistent expression of TROP2 in specimens with invasive features suggests its potential importance in PCa progression and highlights the need for further investigation into its role in lymphovascular invasion and other aggressive cancer behaviors.
When studying the diagnostic performance of TROP2 in discrimination between PCa and BPH, we found that TROP2 exhibited a sensitivity of 100%, which indicates its strong ability to detect malignant prostatic lesions. However, TROP2’s specificity was only 73.33%. The positive predictive value (PPV) and negative predictive value (NPV) for TROP2 were 88.2% and 100% respectively.
TROP2 demonstrated statistically significant validity in differentiating Prostatic Carcinoma from Benign prostatic hyperplasia (p-value < 0.001). The diagnostic strength of TROP2’s sensitivity offers a promising strategy for accurately identifying malignant prostatic lesions. This study is the first to document the diagnostic capability of TROP2 in distinguishing PCa from BPH suggesting that this marker could improve early detection, diagnosis, and prognosis of prostatic carcinoma in clinical practice. In conclusion, TROP2 is a sensitive prostatic immunohistochemical marker and its expression in PCa is higher than that in BPH. TROP2 is associated with poor prognostic factors of PCa as perineural invasion.
So, addition of TROP2 to the immunohistocchemical diagnostic panel of prostatic carcinoma or targeting it in therapy regimens could improve outcome of prostatic carcinoma patients.
It is recommended to further evaluateTROP2 expression in large scale studies with bigger sample size including different histological subtypes of prostatic carcinoma together with follow up of prostatic carcinoma specimens that shows significant TROP2 expression to monitor survival rates and prognosis of patients. This may provide insight into prognostic significance of TROP2 expression in prostatic carcinoma.
Declarations
Availability of data and materials
All data generated or analysed during this study are included in this article.
Competing interests
The authors declare that they have no competing relevant financial or non-financial interests to disclose.
Author contributions
M.A., M.H., M.O., S.F., M.K. contributed to research conception and design, data acquisition, statistical analysis, data analysis, interpretation, drafting and critical revision of the manuscript. M.H., M.O., S.F., M.K. commented on previous versions of the manuscript. M.A., M.H., M.O., S.F., M.K. read and approved the final manuscript.
Ethical approval
The study protocol was reviewed and accepted by the institutional review board and institutional research ethics committee of Faculty of Medicine Suez Canal University prior to initiation (22/3/2023), s.no (5306).
The research involved no risks over researcher, sample or environment.
The need for informed consent has been waived by the institutional review board and institutional research ethics committee of Faculty of Medicine Suez Canal University. The study used human data and material (paraffin blocks) and it was adherent to the declaration of Helsinki and this was insured by the institutional review board and institutional research ethics committee of Faculty of Medicine Suez Canal University.
References
- Prostate cancer Rebello RJ , Oing C, Knudsen KE , Loeb S, Johnson DC , Reiter RE , Gillessen S, Van der Kwast T, Bristow RG . Nature Reviews. Disease Primers.2021;7(1). CrossRef
- Prostate Cancer. Textb Men’s Heal Aging 2 Peyromaure M, Ravery V, Boccon-Gibod L. 2023;:131-146.
- Burden of prostate cancer in the Middle East: A comparative analysis based on global cancer observatory data Kearney G, Chen M, Mula-Hussain L, Skelton M, Eren ME , Orio PF , Nguyen PL , D'Amico AV , Sayan M. Cancer Medicine.2023;12(23). CrossRef
- Global Burden of Prostate Cancer and Association with Socioeconomic Status, 1990-2019: A Systematic Analysis from the Global Burden of Disease Study Zhang W, Cao G, Wu F, Wang Y, Liu Z, Hu H, Xu K. Journal of Epidemiology and Global Health.2023;13(3). CrossRef
- Medicinal plants cultivated in Egypt with anticancer potential; a systematic review Elshnoudy IA , Elkhouly AM , Masoud M, Rabea HA , Mansour FR . Phytochem Rev.2024;2024:1-57.
- Trop2 and its overexpression in cancers: regulation and clinical/therapeutic implications Shvartsur A, Bonavida B. Genes & Cancer.2015;6(3-4). CrossRef
- Trop2: Jack of All Trades, Master of None Lenárt S, Lenárt P, Šmarda J, Remšík J, Souček K, Beneš P. Cancers.2020;12(11). CrossRef
- Targeting Trop-2 in cancer: Recent research progress and clinical application Qiu S, Zhang J, Wang Z, Lan H, Hou J, Zhang N, Wang X, Lu H. Biochimica Et Biophysica Acta. Reviews on Cancer.2023;1878(4). CrossRef
- Increased expression of Trop2 correlates with poor survival in extranodal NK/T cell lymphoma, nasal type Chen R, Lu M, Wang J, Zhang D, Lin H, Zhu H, Zhang W, Xiong L, Ma J, Mao Y, Zhu J, Xu J. Virchows Archiv: An International Journal of Pathology.2013;463(5). CrossRef
- Impact of TROP2 expression on prognosis in solid tumors: A Systematic Review and Meta-analysis Zeng P, Chen M, Zhou L, Tang M, Liu C, Lu P. Scientific Reports.2016;6. CrossRef
- Regulated proteolysis of Trop2 drives epithelial hyperplasia and stem cell self-renewal via β-catenin signaling Stoyanova T, Goldstein AS , Cai H, Drake JM , Huang J, Witte ON . Genes & Development.2012;26(20). CrossRef
- Human prostate sphere-forming cells represent a subset of basal epithelial cells capable of glandular regeneration in vivo Garraway IP , Sun W, Tran CP , Perner S, Zhang B, Goldstein AS , Hahm SA , et al . The Prostate.2010;70(5). CrossRef
- Trop-2 is up-regulated in invasive prostate cancer and displaces FAK from focal contacts Trerotola M, Ganguly KK, Fazli L, Fedele C, Lu H, Dutta A, Liu Q, et al . Oncotarget.2015;6(16). CrossRef
- Trop2 is a driver of metastatic prostate cancer with neuroendocrine phenotype via PARP1 Hsu E, Rice MA , Bermudez A, Marques FJG , Aslan M, Liu S, Ghoochani A, et al . Proceedings of the National Academy of Sciences of the United States of America.2020;117(4). CrossRef
- Could trop-2 overexpression indicate tumor aggressiveness among prostatic adenocarcinomas? Akarken İ, Dere Y. Annals of Diagnostic Pathology.2021;50. CrossRef
- The role of Trop2 in prostate cancer: an oncogene, biomarker, and therapeutic target Shen M, Liu S, Stoyanova T. American Journal of Clinical and Experimental Urology.2021;9(1).
- Overview of Trop-2 in Cancer: From Pre-Clinical Studies to Future Directions in Clinical Settings Lombardi P, Filetti M, Falcone R, Altamura V, Paroni Sterbini F, Bria E, Fabi A, et al . Cancers.2023;15(6). CrossRef
License

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
Copyright
© Asian Pacific Journal of Cancer Biology , 2026
Author Details