Combination Therapy with Hyperthermia, Low-dose Anti-cancer Drug Therapy, and Intravesical Bacillus Calmette–Guérin Instillation for High-risk Non-muscular Invasive Bladder Cancer: A Case of Long-term Survival
Download
Abstract
Introduction: We report a case of long-term survival in a patient with high-risk non-muscular invasive bladder cancer(NMIBC) treated with hyperthermia and low-dose anti-cancer drug therapy combined with intravesical bacillus Calmette–Guérin (BCG) instillation.
Methods:The patient was a male in his 70s. He underwent a left nephroureterectomy for left renal pelvic cancer. Six months later, right renal pelvic cancer was detected. The patient was treated with hyperthermia, radiation therapy, and low-dose anti-cancer drug therapy because he declined surgical treatment. Four years later, he underwent transurethral resection (TUR) of bladder cancer (pathology: urothelial carcinoma (UC) pT1 grade (G) 3) due to recurrence in the bladder. Intravesical instillation of anti-cancer drugs. Next, TUR for urethral recurrence and random bladder biopsy were performed. The pathological diagnosis was UC pT1 G3, and extensive in situ was detected in the bladder. The intravesical instillation of BCG and TUR was conducted four times for urethral recurrence.
Result: During these episodes, the patient refused surgery, and he has been aliving without disturbing his quality of life.
Conclusion: Hyperthermia and low-dose anti-cancer drug treatment combing with intravesical BCG instillation for high-risk non-muscular invasive bladder cancer is effective for the patient refusing standard treatment.
Objective
The ideal goal in treating malignant tumors is to eliminate the tumor from the patient’s body through a combination of surgery, radiation, anti-cancer drugs, and other effective tools. However, despite current advances in medical science, many tumors remain incurable, and the side effects of standard treatments are frequently severe and sometimes cannot be completed in full doses. Therefore, another goal is to maintain malignant tumors in a coexistent and non-aggravating state for as long as possible, even if they are not completely cured. Additionally, a few cases exist where patients prefer not to receive standard treatment.
Here, we report a case of long-term survival in a high-risk patient with NMIBC treated with intravesical BCG instillation followed by hyperthermia and low-dose anti-cancer drugs at our clinic.
Case Presentation
A male patient in his 70s underwent a left nephroureterectomy for left renal pelvis cancer. The patient had no relevant medical history. He smoked 10 cigarettes per day for the past 50 years. Six months later, right renal pelvic cancer was detected. Hyperthermia and extremely low-dose anti-cancer drug therapy were initiated with the hope of avoiding surgery. The first anti-cancer drug administered was cisplatin (CDDP). The anti-cancer drug was changed to mitomycin C (MMC), gemcitabine (GEM), or pirarubicin (THP), as shown in Table 1. Each anti-cancer drug was a single dose determined by his doctor at the time. The dose intensity of MMC was expressed per body since there was no standard dosage set per square meter (/m2). Anti-cancer drugs were administered during hyperthermia therapy (Figure 1 and Table 1).
Figure 1. Details of Treatment Courses.
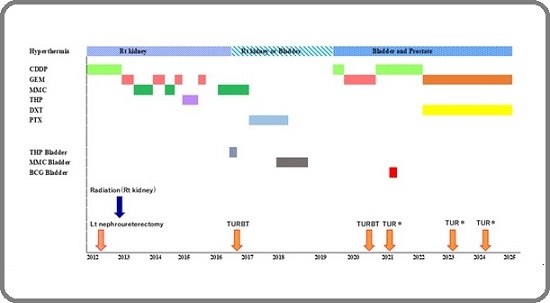
| Hyperthermia | Location | Total nurnber of times | ||
| Right kidney | 217 | |||
| Bladder | 98 | |||
| Prostate: urethra targeted | 109 | |||
| Radiation (Right kidney) | Mass target 3.69 Grayx5f r -10 Grayx5f r=68.45 Gray | |||
| Dilated ureter 3.6 GrayxlOI r=36Gray | ||||
| Chemotherapy | Total nurnber of times | Dose intensicy | Relative dose intensity (%) | |
| CDDP | 2.5 mg | 14 | 127.5 mg/body 1/ week | 2.00 |
| CDDP | 5 mg | 52 | 127.5 mg/body 1/ week | 3.9 |
| CDDP | 10 mg | 42 | 127.5 mg/body 1/ week | 7.80 |
| GEM | 100 mg | 102 | 1700 mg/body 1/ week | 5.90 |
| MMC | 0.1 mg | 65 | 4 mg/body 1/ week | 2.50 |
| MMC | 0.2 mg | 30 | 4 mg/body 1/ week | 5.00 |
| THP | 2 mg | 20 | 11.9 mg/body 1/ week | 16.80 |
| DTX | 5 mg | 62 | 102 mg/body 1/ week | 4.90 |
| PTX | 6 mg | 41 | 119 mg/body 1/ week | 5.00 |
| Bladder instillation | Total nurnber of times | |||
| MMC | 40 mg | 17 | 17 | |
| THP | 30 mg | 5 | 5 | |
| BCG | 40 mg | 8 | 8 |
CDDP: cisplatin, GEM: gemcitabine, MMC: mitomycin C, THP: pirarubicin, DXT: docetaxel, PTX: paclitaxel, BCG: bacillus Calmette–Guérin: fr: frequency. Dose intensity (DI) was determined according to the amounts of anti-cancer drugs based on the lowest dose, and the patient’s surface area was 1.7 m2. Each anti-cancer drug was a single dose determined by his doctor at the time. The DI of MMC was expressed per body since there was no standard dosage set per square meter (/m2). Anti-cancer drugs were administered concurrently with hyperthermia therapy
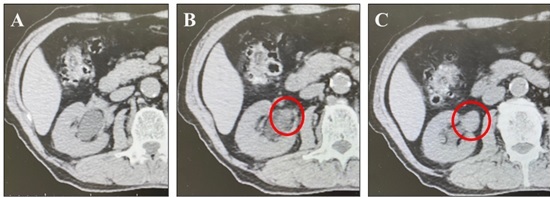
He also underwent radiation therapy for right renal pelvic cancer (Figure. 2A, B, C).
Figure 2. A, The dilated intrarenal pelvis. B, The ureteropelvic junction area is partially occupied with irregular mass (red circle). C: The extrarenal pelvis is filled with a substantial mass (red circle). However, invasion into the surrounding area is not evident. It is judged to be cT3.
Four years later, the patient underwent transurethral resection of bladder cancer (TURBT) due to bladder cancer recurrence. The histopathological diagnosis was urothelial carcinoma (UC) pT1 grade (G) 3, and the diagnosis was pT1cN0cM0. Subsequently, intravesical instillation therapy was initiated with THP. Thereafter, urine cytology remained positive; however, endoscopically, no tumor was found. During this period, hyperthermia was continued in the right kidney and bladder. THP was changed to MMC, intravesical instillation was continued, and urine cytology was negative. At that point, the right renal pelvic cancer disappeared on computed tomography imaging and was diagnosed as a complete response.
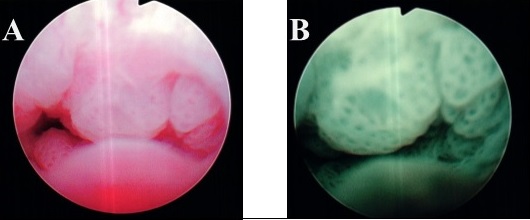
A second TUR for urethral recurrence and a random biopsy of the bladder using photodynamic diagnosis were performed (Figure 3A, B).
Figure 3. A, White Color View of the Urethral Recurrent Tumor. Several stalked, broad-based papillary tumors protruding into the urethra are seen. B, Photodynamic diagnosis (PDD) view of the same tumor. The papillary pattern is clearer than the white color view.
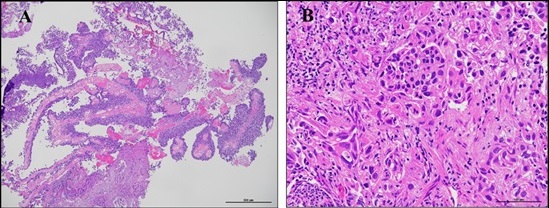
The pathological diagnosis was UC pT1 G3 (Figure 4A, B), and extensive CIS was detected in the bladder. Again, the diagnosis was pT1cN0cM0.
Figure 4. The Urethral Pathological Diagnosis was Urothelial Carcinoma (UC) pT1 G3. A, Papillary stalked tumors are the majority (in the low-power field, × 40, hematoxylin and eosin staining). B, Atypical urothelium with severe nuclear atypia proliferates in a papillary and follicular fashion (in the high-power field, × 200, hematoxylin and eosin staining).
Subsequently, weekly intravesical instillations of BCG were administered eight times. During these periods, the patient received hyperthermia treatment targeting the prostatic urethral region, combined with low-dose anti-cancer drugs, as shown in Table 1, and underwent TUR for urethral recurrence four times. The patient refused surgery despite the persuasion of several physicians and is currently undergoing continuous hyperthermia and low-dose anti-cancer drug treatments.
Discussion
This is the only case of this type presenting in our clinic; however, we confirm that the patient is still alive without invasion or distant metastasis since the diagnosis of left renal pelvis cancer 12 years ago. At our clinic, low-aggressive cancer treatment has been continued according to the patient’s wishes.
The first diagnosed cancer of the renal pelvis was surgically removed; however, the cancer of the opposite renal pelvis could not be removed without hemodialysis. The patient refused surgery; however, after consulting with his doctor, the patient was cured radically with low-dose anti-cancer drugs, hyperthermia, and radiotherapy.
Urothelial cancer is prone to recurrence [1, 2], and the prognosis for renal pelvic cancer is generally considered poorer than that of bladder cancer. Additionally, recurrence in the bladder is common [3]. In this case, the recurrence occurred in the bladder; therefore, TURBT and intravesical instillation of anti-cancer drugs were performed continuously. Eight years after the initial treatment, the patient experienced urethral recurrence combined with extensive CIS. Bladder CIS has a high potential for invasion or distant metastasis, and BCG is highly effective against CIS [4, 5].
During this period, low-dose anti-cancer drugs and hyperthermia were continued, targeting both the right kidney and bladder. In this high-risk NMIBC case, he could continue these treatments while preserving his remaining kidney and bladder, which are indicated to have a poor prognosis.
Hyperthermia therapy has a long history [6-12]. In 1995, Tanaka reported the significance and role of hyperthermia in combination with radiation therapy [13], and in 2008, he reported its usefulness in the Hyperthermia Cancer Thermotherapy Guidebook [14]. Furthermore, in the same guidebook, Imada reported that concomitant therapy with low doses of anti-cancer agents had fewer side effects and maintained efficacy. However, he emphasized that this approach is most effective in localized disease [15].
As mentioned in the introduction section, hyperthermia has long been the subject of basic research, and in Japan, researchers such as Ito et al. and Hasegawa et al. have been active in elucidating its mechanism [7-9]. It is a treatment option that is expected to be effective while reducing the burden on the patient, and in this case, it appears to have been the ideal treatment choice, genuinely aligning with the patient’s needs.
Regarding the lifestyle quality of patients with bladder cancer, stoma creation and dialysis owing to urinary tract changes are burdensome [16]. This is a great advantage for outpatient treatment facilities, such as ours, that lack an inpatient ward, allowing for low-aggressive treatment with reduced patient burden and scheduling according to the patient’s preference. Consequently, our patient could be treated per his preference.
Many patients wish to avoid anti-cancer drug regimens and immuno-oncology drugs that may cause unpredictable side effects. Therefore, there is an ever-increasing number of patients requesting so-called low-aggressive therapies. We sincerely hope that the number of such facilities will increase, thereby promoting the use of hyperthermia therapy and expanding the environment where patients can receive less aggressive treatment. A tailor-made treatment that meets the patient’s needs is desirable.
In conclusion, hyperthermia and low-dose anti-cancer drug treatment combing with intravesical bacillus Calmette–Guérin instillation for high-risk non-muscular invasive bladder cancer is effective for the patient refusing standard treatment.
Conflict of Interest Disclosure
The authors declare no conflict of interest.
Originality Declaration for Figures
All figures included in this manuscript are original and have been created by the authors specifically for the purposes of this study. No previously published or copyrighted images have been used. The authors confirm that all graphical elements, illustrations, and visual materials were generated from the data obtained in the course of this research or designed uniquely for this manuscript.
Ethics approval
This study was approved by the Ethic Committee of the 20251229 Medoc Hearth Clinic.
Consent to participate
Written informed consent was obtained from all participants, and the trial was conducted in accordance with the Declaration of Helsinki.
References
- Recurrence mechanisms of non-muscle-invasive bladder cancer - a clinical perspective Teoh JY , Kamat AM , Black PC , Grivas P, Shariat SF , Babjuk M. Nature Reviews. Urology.2022;19(5). CrossRef
- Urothelial carcinoma: Recurrence and risk factors Milojevic B, Dzamic Z, Kajmakovic B, Milenkovic Petronic D, Sipetic Grujicic S. Journal of B.U.ON.: official journal of the Balkan Union of Oncology.2015;20(2). CrossRef
- European association of urology guidelines on upper urinary tract urothelial carcinoma: 2020 update Roupr^et M, Burger M, Capoun O, Cohen D, Compe´ rat EM , Cowan NC , et al . Eur Urol.2021;79(1). CrossRef
- Diagnosis and management of urothelial carcinoma in situ of the lower urinary tract: a systematic review Casey RG , Catto JWF , Cheng L , Cookson MS , Harry Herr H , Shariat S , Witjes JA , Black PC . Eur Urol.2015;67(5). CrossRef
- Bladder cancer: a review Lenis AT , Lec PM , Chamie K , Mshs MD . JAMA.2020;324(19). CrossRef
- Selective inductive heating of lymph nodes Gilchrist RK , Medal R , Shorey WD , Hanselman RC , Parrott JC , Taylor CB . Ann Surg.1957;146(4). CrossRef
- Combining antitumor drugs with mild hyperthermia increases the cytotoxic effects of drugs on human leukemia cells in vitro Itoh Y , Kazaoka Y , Nitta M , Yamada Y , Honda N . Mol Med Rep.2009;2(3). CrossRef
- Mild-hyperthermia enhances tumor drug uptake and anti-tumor effect when combined with cisplatin as chemotherapy Hasegawa T, Takahashi T, Uno K. PASKEN J.2019;26-29:40-49.
- Hyperthermic immuno-cellular therapy-basic and clinical study Takeda T, Fukunaga K, Miyazawa K, Takahashi T, Takeda H, Takeda Y, Tanigawa K, et al . Gan to Kagaku Ryoho. Cancer & Chemotherapy.2008;35(12).
- Pathological complete response and sphincter-sparing surgery after neoadjuvant radiochemotherapy with regional hyperthermia for locally advanced rectal cancer compared with radiochemotherapy alone Schroeder C , Gani C , Lamprecht U , Weyhern CH , Weinmann M , Bamberg M , Berger B . Int J Hyperth.2012. CrossRef
- Wust P. Radiochemotherapy combined with regional pelvic hyperthermia induces high response and resectability rates in patients with nonresectable cervical cancer > or = FIGO IIB “bulky” Sreenivasa G , Hildebrandt B , Kummel S , Jungnickel K , Cho CH , Tilly W , Böhmer D , et al . Int J Radiat Oncol Biol Phys.2006;66(4). CrossRef
- Effect of concurrent chemotherapy and hyperthermia on outcome of preoperative radiotherapy of high-risk soft tissue sarcomas Eckert F , Gani C , Kluba T , Mayer F , Kopp HG , Zips D , Bamberg M , Müller AC . Strahlenther Onkol.2013;189(6). CrossRef
- Significance and role of hyperthermia in multidisciplinary treatment Tanaka R. Jpn J Hypertherm Oncol.1995;11:137-48.
- Hyperthermia - Cancer Hyperthermia Guidebook. 1.03 Combination with radiation. Jpn Soc Therm Med. 2008 8-11. 15. Imada H. Hyperthermia - Cancer Hyperthermia Guidebook. 1.04 Combination with anti-cancer agents Tanaka R. Jpn Soc Therm Med.2008;:12-13.
- Hyperthermia - Cancer Hyperthermia Guidebook. 1.04 Combination with anti-cancer agents Imada H. Jpn Soc Therm Med.2008;:12-13.
- Lifestyle factors and health-related quality of life in bladder cancer survivors: a systematic review Gopalakrishna A, Longo TA , Fantony JJ , Van Noord M, Inman BA . Journal of Cancer Survivorship: Research and Practice.2016;10(5). CrossRef
License

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
Copyright
© Asian Pacific Journal of Cancer Care , 2026
Author Details
How to Cite
- Abstract viewed - 0 times
- PDF (FULL TEXT) downloaded - 0 times
- XML downloaded - 0 times